Spinal cord injury (SCI) represents a formidable challenge in the field of neural repair. Once it occurs, it often leads to irreversible sequelae such as paralysis and sensory loss, profoundly impacting patients' quality of life. The core obstacles lie in the extremely limited regenerative capacity of the central nervous system and the inhibitory microenvironment that forms post-injury, acting as an "invisible barrier" that impedes axonal regeneration and functional remodelling. Consequently, developing novel therapeutic strategies capable of replacing damaged neural tissue, reconstructing neural circuits, and restoring limb function has become a critical goal for research teams worldwide.
On 19th December 2025, a research team led by Shao Zhicheng from the Institute of Translational Brain Research, Fudan University, in collaboration with Professor Wu Shengxi from the Air Force Medical University, published a study entitled "Transplantation of engineered spinal cord organoids restores functions after spinal cord injury" in the prestigious international journal Brain, marking a significant breakthrough in this area. This study not only provides the first confirmation of the feasibility and efficacy of using engineered scaffold-supported spinal cord organoids for repairing complete spinal cord injury but also establishes a crucial theoretical foundation and technological reserve for the future development of "artificial spinal cord tissue" for clinical application. Furthermore, the large-scale organoid model with complex structure and function developed in this research offers an important experimental platform for studying neural development, mechanisms of spinal cord diseases, and drug screening, thereby advancing the fields of neural regenerative medicine and organoid technology.

The team innovatively constructed a nano-scaffold system featuring a biomimetic vascular network structure by combining carboxylated cellulose nanofibers (CCN) with Matrigel. This CCN-Matrigel biomimetic scaffold not only supported the normal developmental trajectory of the organoids but also significantly enhanced their viability. The scaffold structure substantially improved the efficiency of nutrient exchange within the organoids, leading to a marked reduction in the expression of apoptosis-related genes and effectively preventing central necrosis (Figure 1).
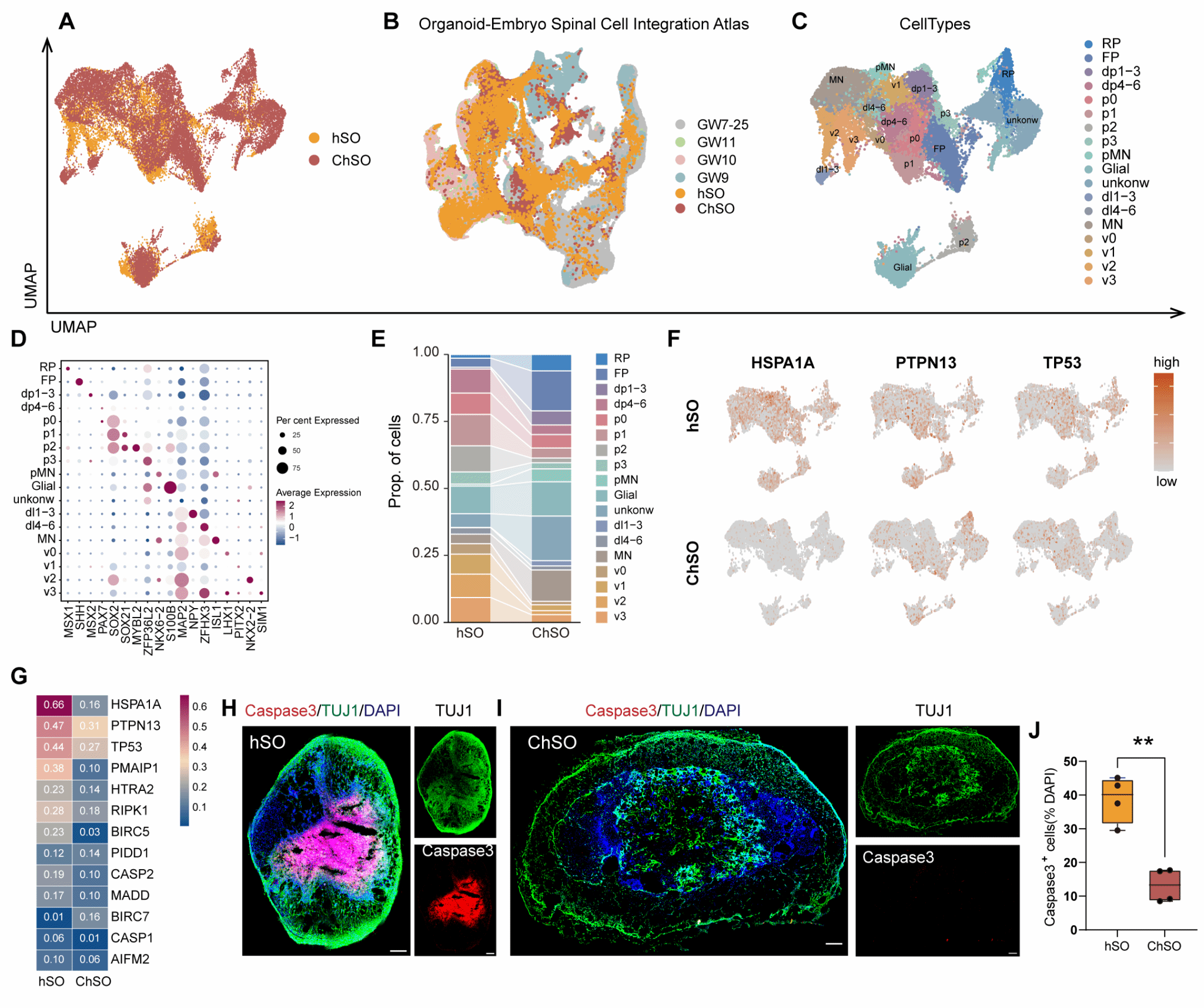
Figure 1. CCN-Matrigel scaffold maintains layer-specific differentiation of spinal cord organoids while effectively preventing central necrosis
Long-term cultured engineered spinal cord organoids (ChSOs) exhibited remarkable structural maturation and functional integration capabilities. They spontaneously extended aligned axon bundles and achieved myelination ensheathing the axons, recapitulating the conductive structure of neural fibres in vivo. Building on this foundation, these organoids further developed into neural networks exhibiting synchronous electrical activity, demonstrating complex spontaneous firing, synaptic transmission, and even rhythmic oscillations. This successfully recreated key electrophysiological functions of mature spinal cord tissue, providing a highly biomimetic in vitro model for studying neural circuit development and injury repair (Figure 2).
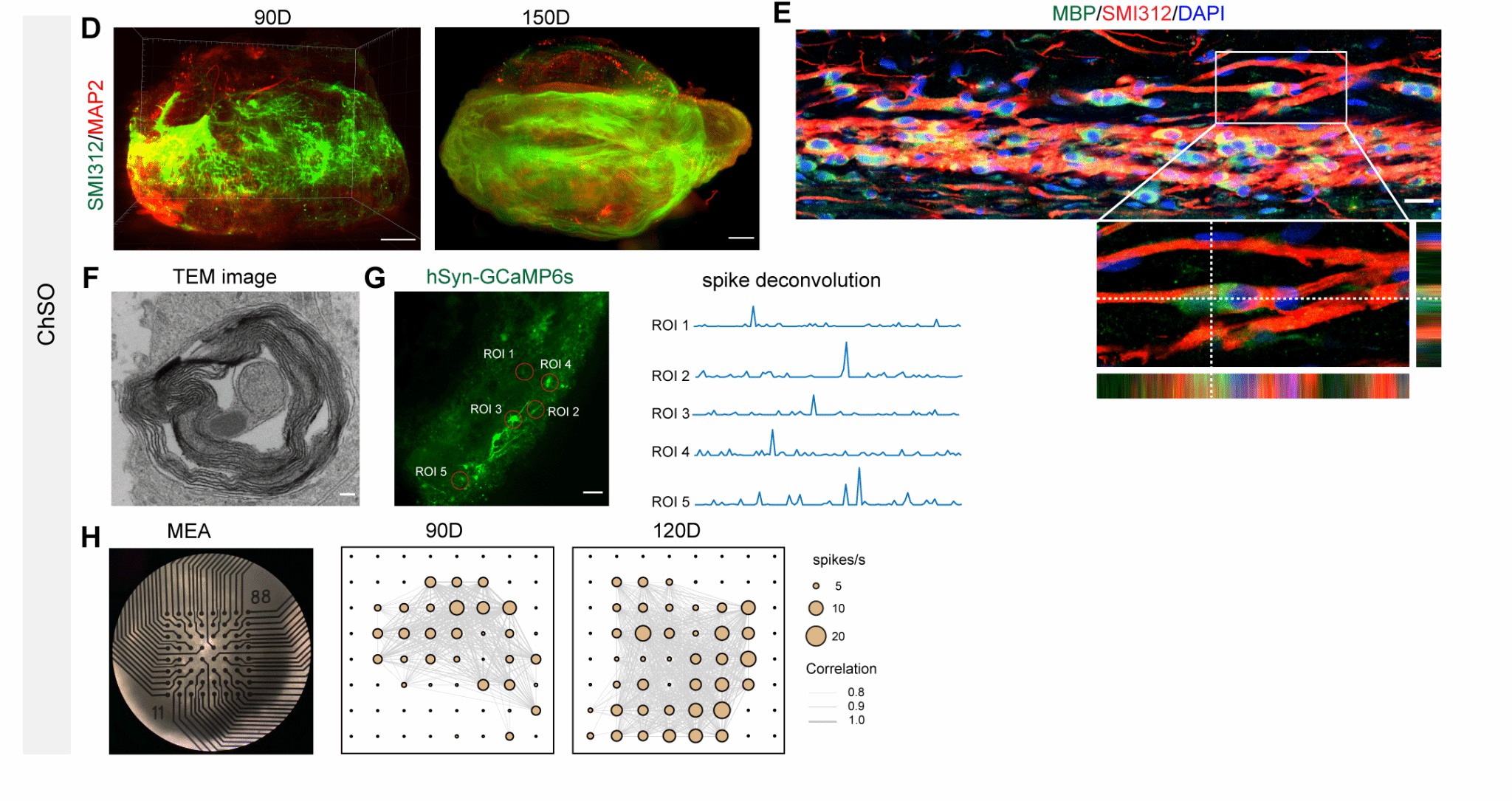
Figure 2. Long-term cultured engineered spinal cord organoids exhibit more mature neuronal function and form axon bundles and myelin sheaths
The research team proceeded to transplant the mature engineered spinal cord organoids (ChSOs) into a mouse model of complete spinal cord injury. Histological analysis revealed that the transplanted ChSOs exhibited excellent survival and structural integration within the injured area. They not only differentiated into various spinal neuron types with distinct dorsal-ventral characteristics, including motor neurons and excitatory/inhibitory interneurons, but also generated astrocytes and myelinating oligodendrocytes. These results demonstrate that ChSOs retain their multi-lineage differentiation potential within the in vivo environment and can form, together with host tissue, a complex neural microenvironment comprising neurons and supporting cells, establishing a cellular foundation for functional repair (Figure 3).
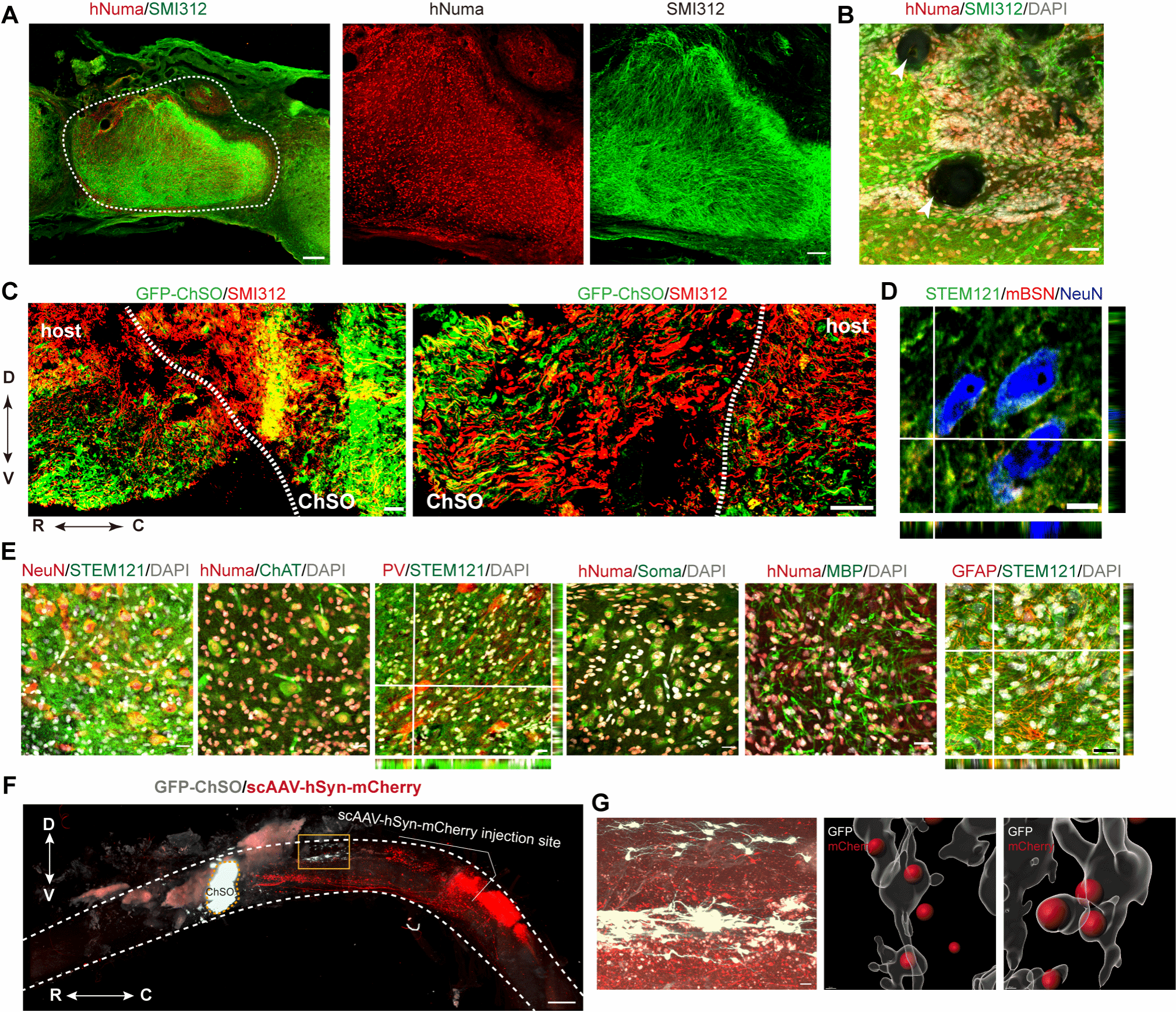
Figure 3. Survival and neuronal differentiation of transplanted ChSOs in mice with complete SCI
Over a 12-week long-term behavioural assessment, mice receiving ChSO transplants showed significant and sustained neurological functional recovery. Quantified using the Basso Mouse Scale scoring system, the motor function scores of the hindlimbs continuously improved, significantly surpassing those of control groups that received no transplant or scaffold-only transplants. Tactile testing revealed a significantly reduced paw withdrawal threshold in the transplant group, indicating effective restoration of sensory perception. Notably, some mice even regained alternating gait and weight-bearing capacity in their hindlimbs, reflecting improved higher-order motor coordination. Collectively, these behavioural findings confirm that transplanted ChSOs can promote sensorimotor functional reconstruction following complete spinal cord injury (Figure 4).
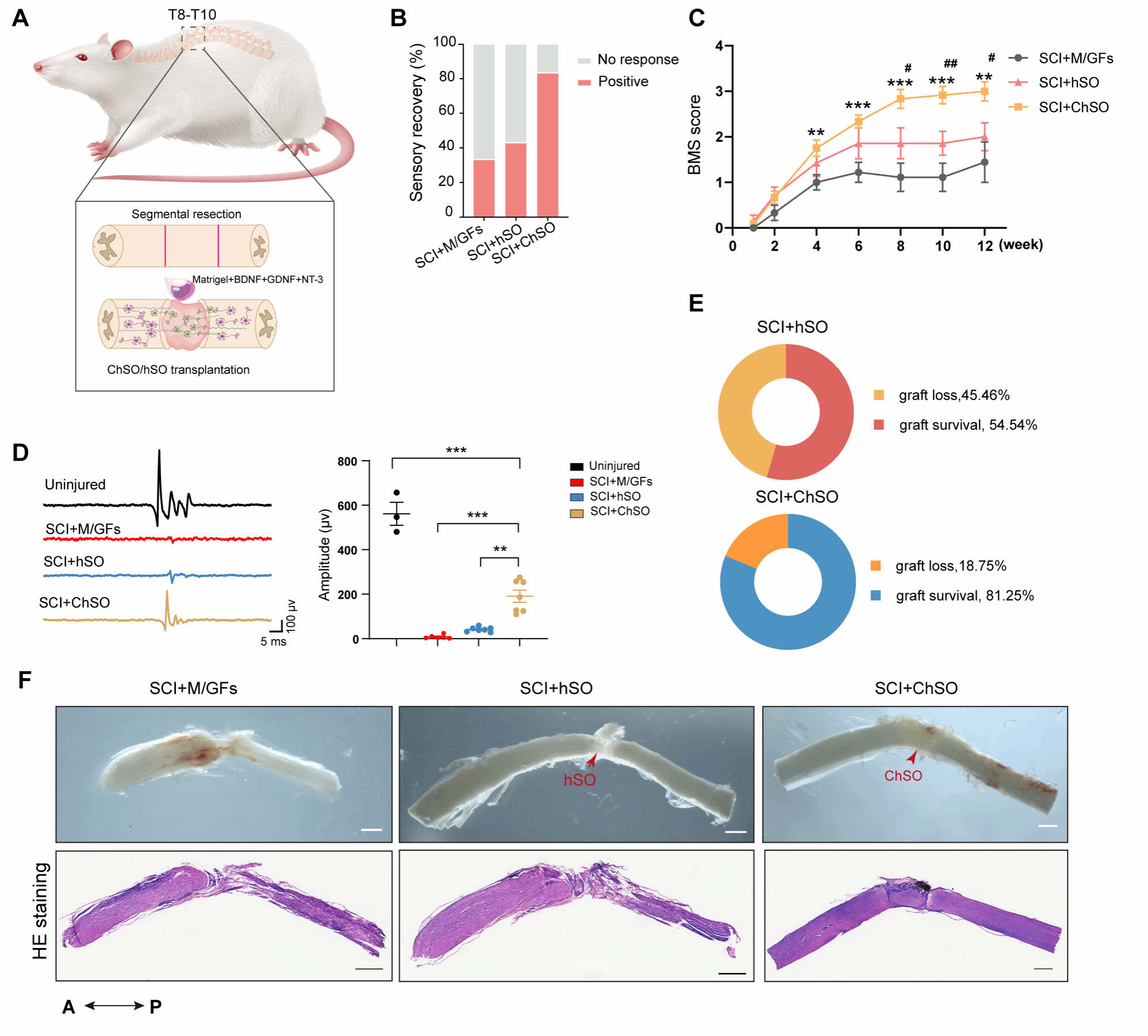
Figure 4. Comparison of survival and functional recovery effects between engineered spinal cord organoids (ChSOs) and traditional spinal cord organoids (hSOs) in a complete SCI model
In summary, the large-scale human spinal cord organoids successfully constructed by this team not only open up a novel therapeutic avenue for spinal cord regeneration research but also provide a highly adaptable and translationally relevant model for studying spinal cord-related diseases. This breakthrough significantly advances the development and translational application of organoid technology in the field of central nervous system regeneration, laying an important foundation for innovation in future clinical treatment strategies.
Researcher Shao Zhicheng from the Institute for Translational Brain Research, Fudan University, and Professor Wu Shengxi from the Air Force Medical University are the co-corresponding authors of this paper. Dr Liu Linlin, a postdoctoral fellow, and Associate Researcher Xue Weiwei, both from the Institute for Translational Brain Research, are the co-first authors. This research was supported by grants from the National Natural Science Foundation of China Original Exploration Project, the National Key Research and Development Program of China, and the Yangtze River Delta Joint Innovation and Research Project.
Link to the original article: https://academic.oup.com/brain/advance-article/doi/10.1093/brain/awaf471/8384044?login=true