The sympathetic nervous system is widely recognised for its role in orchestrating the "fight or flight" response during life-threatening situations. However, accumulating evidence suggests that the sympathetic nervous system also plays a crucial part in fine-tuning the physiological state of organs to maintain internal homeostasis. This implies a specific functional division of labour among sympathetic neural circuits, conceptualised as sympathetic functional units. However, the precise organisational principles of these functional units, along with their cellular and circuit basis, have remained a central unresolved question in autonomic neuroscience.

On 10th December 2025, research groups led by Ni Jinfei, Duan Shumin, and Tan Chao at the Institute for Translational Brain Research, Fudan University, collaboratively published a study online in Neuron entitled "Sympathetic functional units encoded by genetically defined postganglionic neurons". Using the coeliac-superior mesenteric ganglion complex (CG-SMG) as a model system, this research systematically explored the organisational principles of sympathetic functional units. For the first time, it revealed that the functions regulating gastrointestinal motility and visceral vasoconstriction are carried out by two distinct populations of sympathetic postganglionic neurons. These two neuronal populations exhibit marked differences in both gene expression and terminal morphology. This discovery not only provides the first evidence at the cellular level confirming the existence of distinct sympathetic functional units but also offers a critical breakthrough for further understanding how the brain achieves precise regulation of organ function through the combinatorial action of such functional units.
Previous research on sympathetic functional units largely relied on earlier physiological and clinical experiments. Whilst these studies proposed that the sympathetic nervous system might possess multiple functional modules for finely regulating different physiological processes, this work remained largely conceptual. No studies had definitively identified which specific neurons constitute various sympathetic functional units, nor had they characterised their organisational structure and functional features. In this study, we employed a multi-omics approach, combining anatomical tracing, single-cell transcriptomics, and cell-type-specific functional experiments to systematically dissect the postganglionic sympathetic neurons within the coeliac-superior mesenteric ganglion complex (CG-SMG).
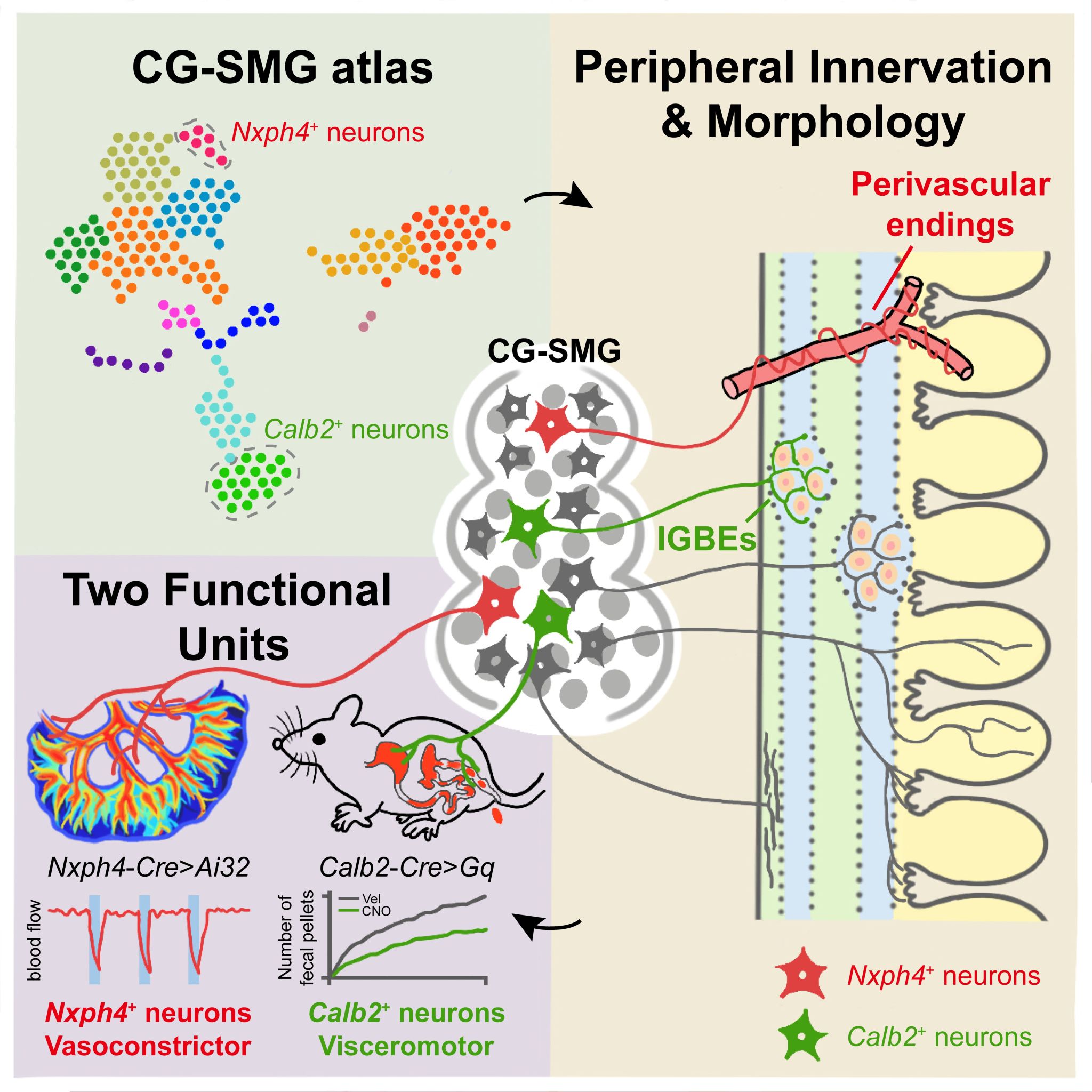
The CG-SMG contains sympathetic postganglionic neurons that provide sympathetic innervation to various abdominal organs, forming diverse types of nerve endings. Notably, in the projection map of the gastrointestinal tract, we found that the CG-SMG projects to both the myenteric and submucosal layers of the gut, exhibiting multiple types of terminal endings: IMAs, IGBEs, perivascular endings, and villi endings. Different CG-SMG nerve terminal types may correspond to the regulation of distinct physiological functions (Figure 1).
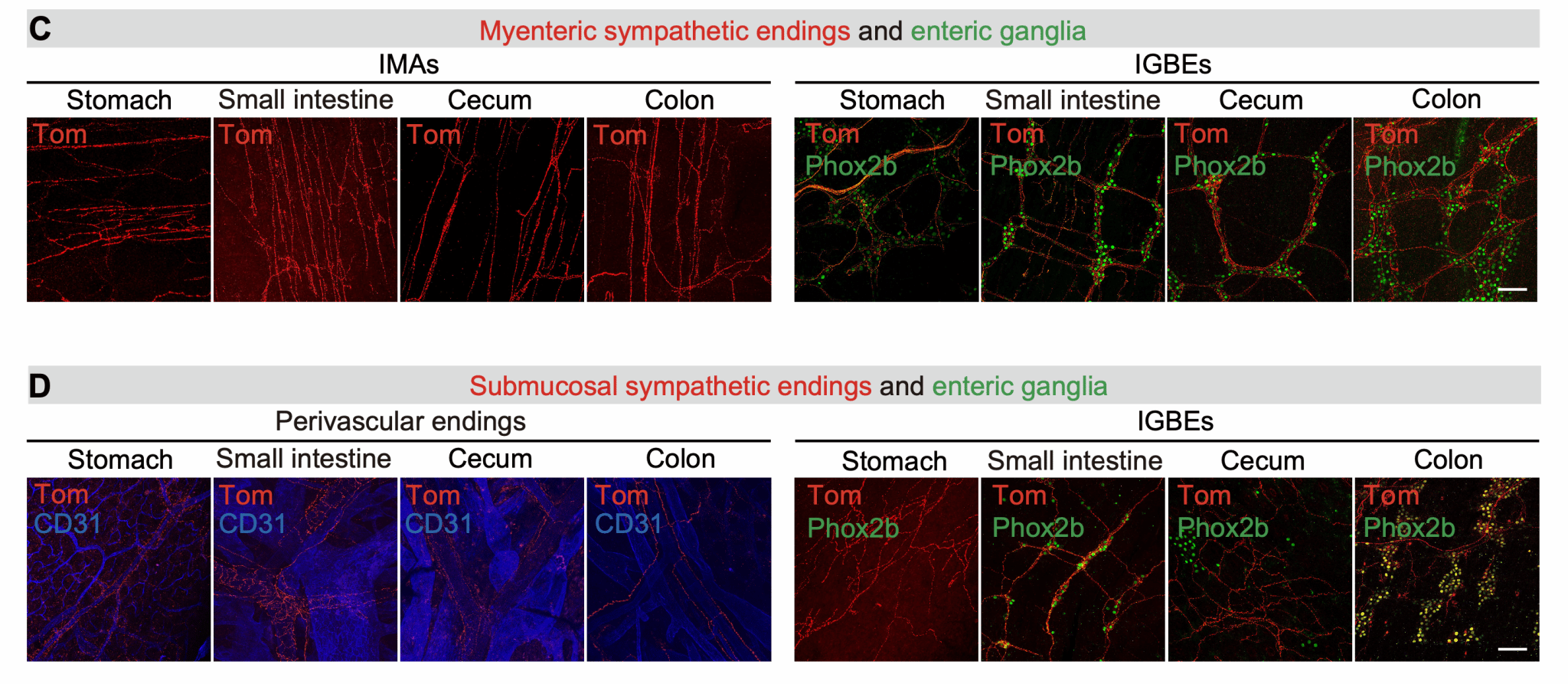
Figure 1. CG-SMG sympathetic neurons innervate the gastrointestinal tract through morphologically distinct nerve terminal structures
To further elucidate the cellular and molecular diversity of CG-SMG postganglionic sympathetic neurons, we performed single-cell transcriptomic analysis. This revealed that the CG-SMG can be divided into multiple neuronal subtypes, describing a previously unrecognised molecular diversity among CG-SMG sympathetic neurons (Figure 2). Based on the differentially expressed genes of each subtype, we generated and collected various Cre mouse lines. Utilising these Cre mouse lines enabled us to label and manipulate different neuronal subtypes within the CG-SMG.
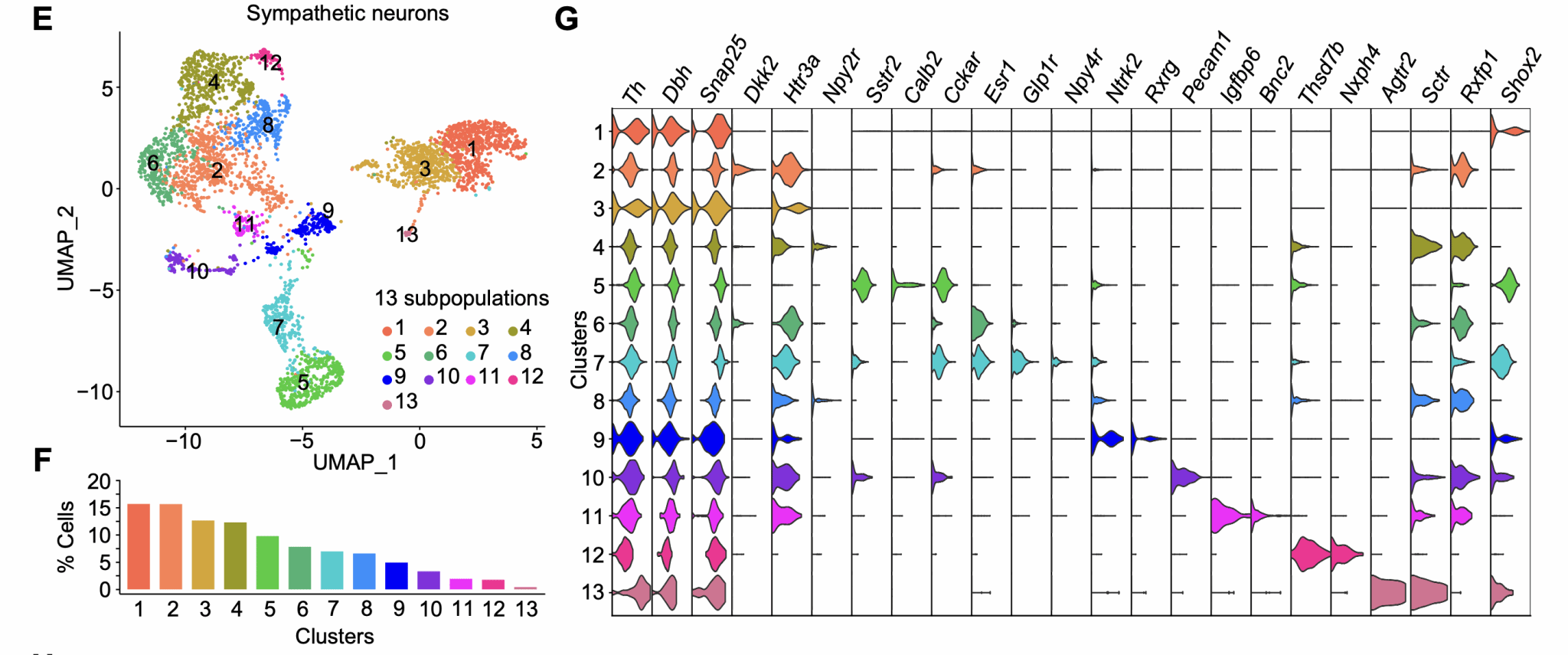
Figure 2. Single-cell transcriptomics reveals cellular diversity of CG-SMG sympathetic neurons
If different sympathetic functional units are indeed composed of distinct CG-SMG neuronal subtypes, then further analysis of the tracing and physiological function of each subtype was warranted. Fortuitously, we identified two non-overlapping neuronal subpopulations within the CG-SMG: Calb2-positive neurons and Nxph4-positive neurons. These possess specific projection characteristics and correspond to the regulation of different physiological functions.
Specifically, the Calb2-positive neuronal subtype within the CG-SMG projects exclusively to the myenteric plexus layer of the gastrointestinal tract, with no projections to the submucosal layer or other abdominal organs. Furthermore, the terminals they form within the gut myenteric layer are exclusively those surrounding myenteric plexus neurons (IGBEs). This suggested that Calb2-positive neurons might constitute a "motility functional unit" neuronal population. Subsequent physiological functional experiments confirmed this: activating Calb2-positive neurons in the CG-SMG significantly increased gastrointestinal transit time and markedly reduced faecal pellet output. In contrast, activating Nxph4-positive neurons did not alter the mice's gastrointestinal motility status. Through projection mapping and physiological functional analysis, Calb2-positive neurons were indeed confirmed as "motility functional unit" neurons (Figure 3).
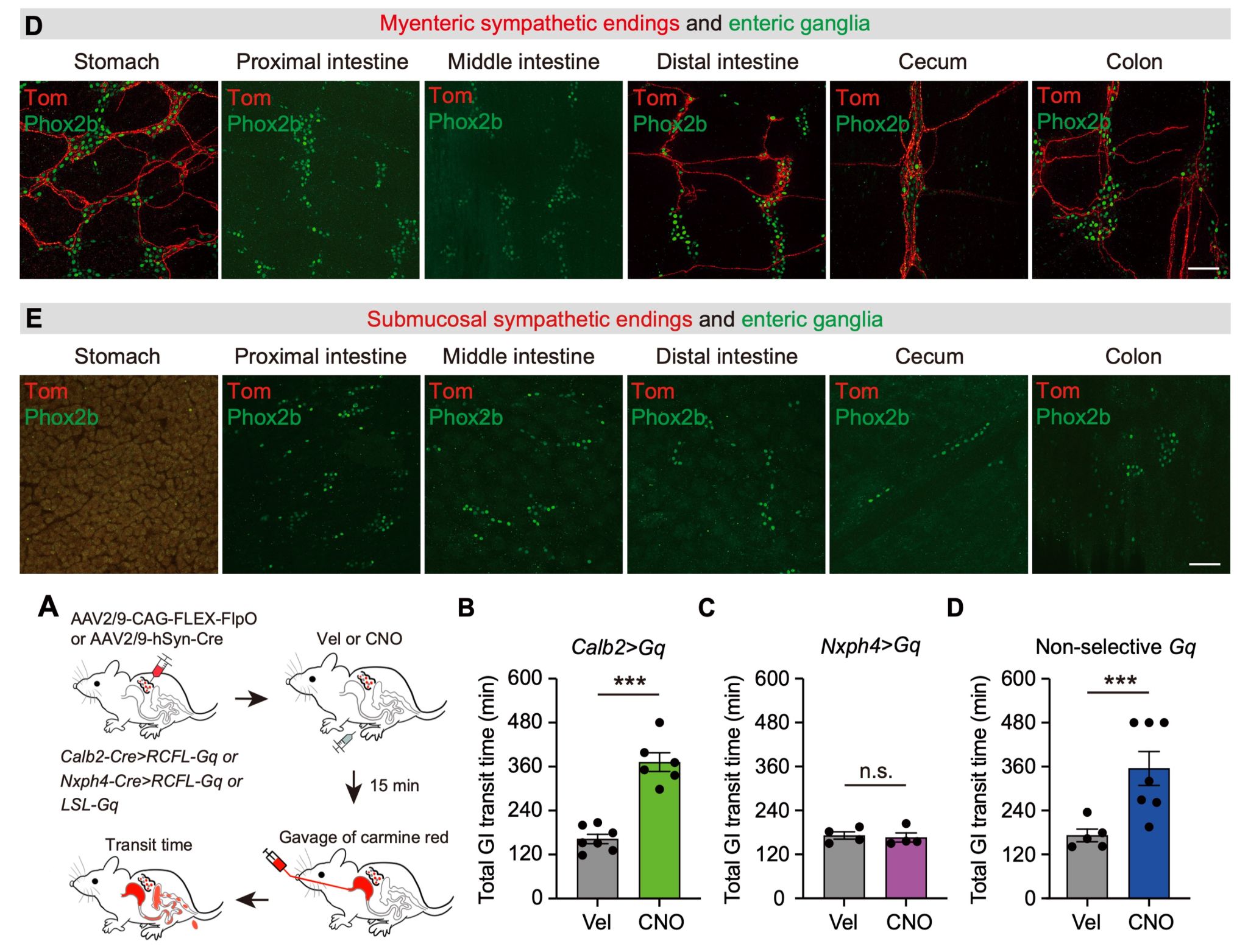
Figure 3. Calb2-positive neurons in the CG-SMG represent "motility functional unit" neurons
Analysis of the projection patterns of Nxph4-positive neurons in the CG-SMG revealed that they form perivascular endings around blood vessels in both the gastrointestinal tract and other abdominal organs. Based on this, we hypothesised that Nxph4-positive neurons might play an important regulatory role in controlling vasoconstriction and vasodilation. Further optogenetic activation of Nxph4-positive neurons in the CG-SMG resulted in a marked constriction of intestinal blood vessels and a significant decrease in blood perfusion during light stimulation. Conversely, optogenetic activation of Calb2-positive neurons did not affect blood flow. These results confirmed that Nxph4-positive neurons constitute a "vasoconstriction functional unit" neuronal population (Figure 4).
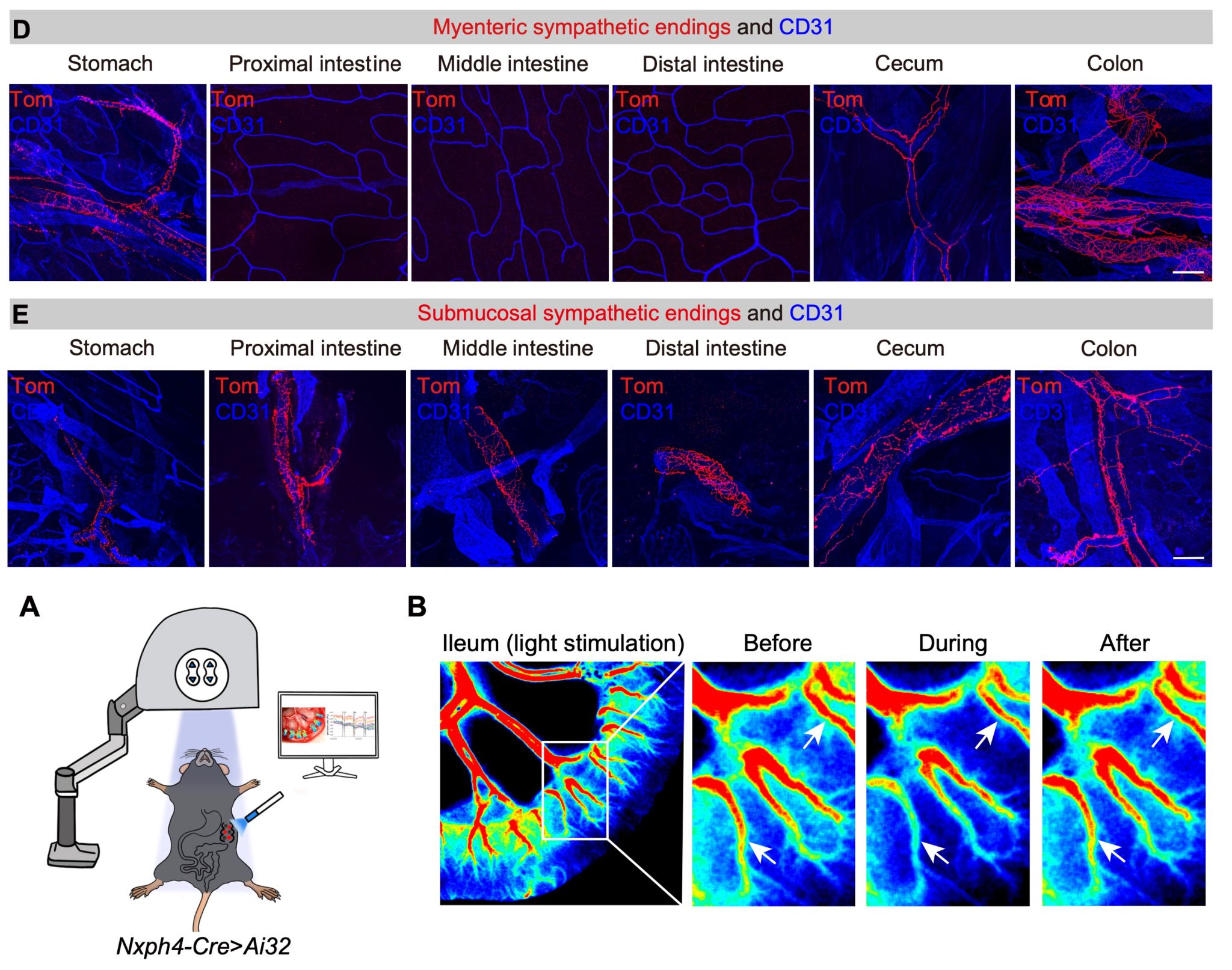
Figure 4. Nxph4-positive neurons in the CG-SMG represent "vasoconstriction functional unit" neurons
This study has, for the first time, clarified the organisational principles of sympathetic functional units, demonstrating that postganglionic sympathetic neurons in the sympathetic nervous system comprise two molecularly defined functional unit types: Calb2-positive neurons as "motility functional unit" neurons and Nxph4-positive neurons as "vasoconstriction functional unit" neurons. It resolves a key question in the sympathetic field: different sympathetic functional units are composed of distinct postganglionic neuronal subtypes. This finding suggests that sympathetic functional circuits employ a form of "labelled line" coding to achieve functional regulation of target organs. By identifying these postganglionic neurons of sympathetic functional units, we can now begin to uncover the circuit mechanisms through which the brain achieves fine-tuned control of organ physiology via the brain-body axis and the sympathetic system. This research elevates the field's understanding of this fundamental question concerning sympathetic functional units to a new dimension.
Researcher Ni Jinfei, Academician Duan Shumin, and Researcher Tan Chao from the Institute for Translational Brain Research, Fudan University, are the co-corresponding authors of this paper. Dr Wei Yaru and doctoral student Ma Zongya, both from the same institute, are the co-first authors. This research was supported by funding from the 2021 Science and Technology Innovation 2030 "Brain Science and Brain-Inspired Research" Major Project, the National Natural Science Foundation of China, and the China Postdoctoral Science Foundation.
Link to the original article: https://www.cell.com/neuron/fulltext/S0896-6273(25)00809-8
Dr Ni Jinfei's research group is dedicated to studying the autonomic nervous system, including brain-body interactions mediated by interoceptive systems, with a current focus on the structure and function of neural connections between digestive system organs and the brain. Scientists interested in these questions are welcome to discuss potential collaborations.

WeChat ID | Jinfei Ni
Welcome for discussions and collaboration