Chronic stress-related mood disorders, such as anxiety and depression, represent a group of highly prevalent mental health conditions globally. The pathogenesis of these disorders is complex, involving multiple levels of dysfunction, including imbalances in neuronal excitability and aberrant neural circuit function. Voltage-gated sodium channels (NaV), core molecules governing the initiation and propagation of neuronal action potentials, have subtypes whose dysfunction is closely linked to mood disorders. Among these, the tetrodotoxin (TTX)-resistant sodium channel NaV1.5 (encoded by the Scn5a gene) has been extensively studied in peripheral cardiac electrophysiology and myocardial function. However, its expression pattern and function within the brain remain unclear. Previous in situ hybridisation experiments suggested that Scn5a is also expressed in brain regions critical for emotional regulation, such as the limbic system. Nevertheless, further investigation was required to determine how it influences neuronal excitability, whether it participates in regulating emotional behaviour, and if it could serve as an ion channel target for intervening in negative emotions.

On 4th February 2026, Professor Shu You-Sheng's research group at the Institute of Translational Brain Research, Fudan University, published a study in Molecular Psychiatry entitled "Tetrodotoxin-resistant NaV1.5 channels regulate excitability of lateral septum neurons and emotion behaviours of chronically stressed mice". This research revealed that NaV1.5, traditionally considered primarily expressed in the heart, is abundantly expressed in neurons of the lateral septum (LS) and plays a crucial role in regulating neuronal excitability and negative emotions.
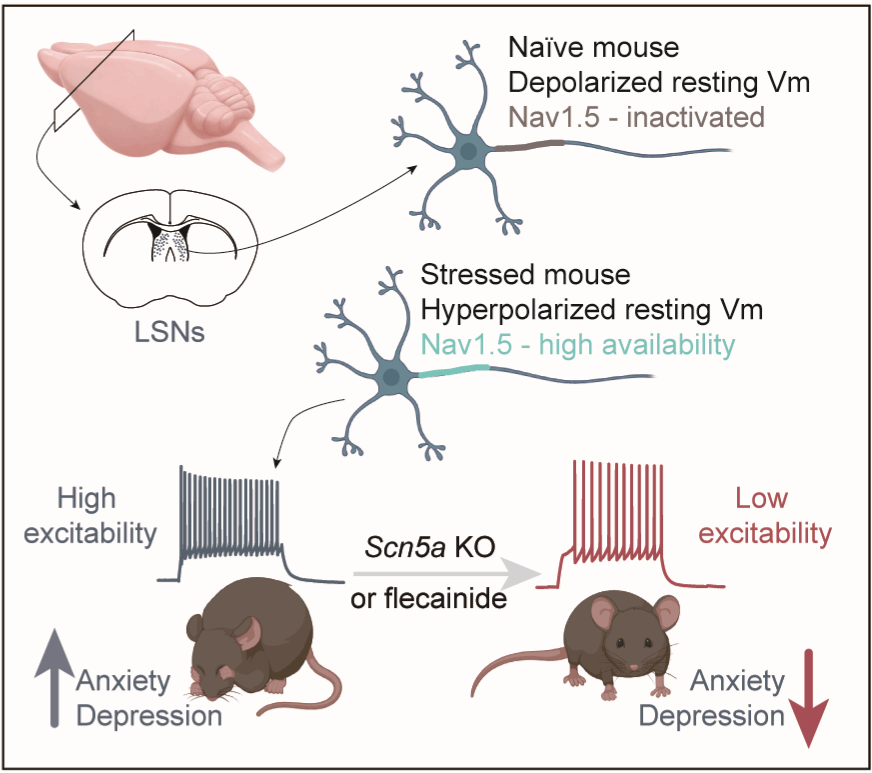
The research team initially employed single-nucleus RNA sequencing combined with immunofluorescence staining and discovered that Scn5a is expressed at high levels across multiple LS neuronal types. Furthermore, the NaV1.5 channel protein was found to be densely clustered in the axon initial segment (the axonal site where action potentials are first generated). This observation suggested that NaV1.5 might directly participate in regulating action potential firing in LS neurons. Subsequent electrophysiological recordings in acute brain slices revealed that NaV1.5 possesses distinctive channel activation properties: when the neuron resides in a hyperpolarised membrane potential state, activating NaV1.5 alone sufficed to trigger action potential generation. This characteristic enables LS neurons to respond rapidly to incoming stimuli from a resting state, indicating NaV1.5's involvement in swift information processing.
Intriguingly, mice with a specific knockout of NaV1.5 in LS neurons did not exhibit significant alterations in emotional behaviour under baseline, non-stressed conditions. However, following exposure to chronic restraint stress, these mice displayed a marked reduction in LS neuronal excitability and demonstrated significantly lower levels of anxiety- and depressive-like behaviours. Specifically, they spent more time in the open arms of the elevated plus maze and showed reduced immobility time in the forced swim test. This finding carries significant clinical implications, suggesting NaV1.5 may represent a "stress-dependent" therapeutic target. Drug intervention targeting this channel could potentially yield therapeutic effects specifically under pathological conditions like chronic stress, without affecting emotional behaviour in the normal state.
Building upon the known pharmacological profile of NaV1.5 in the heart, the team tested the effects of the antiarrhythmic drug flecainide on NaV1.5 currents in LS neurons. Results showed that acute application of flecainide significantly suppressed the TTX-resistant sodium current. Crucially, in chronically stressed mice, a single intraperitoneal injection of flecainide rapidly alleviated anxiety- and depressive-like behaviours. This discovery opens new possibilities for acute intervention in mood disorders. It is important to note that the core conclusions of this study derive from mouse models, and flecainide is indeed known to induce electrocardiographic changes. Its efficacy in patients with mood disorders, along with optimal administration routes, dosages, and long-term safety, remain to be investigated and validated through clinical trials. Furthermore, whether the role of NaV1.5 differs across various subtypes of mood disorders, such as major depressive disorder or social anxiety, warrants further exploration.
In summary, this study elucidates the significant role of NaV1.5 channels in regulating LS neuronal excitability and negative emotions, establishing a novel link between the brain sodium channel NaV1.5 and emotional control. These findings not only broaden our understanding of the neural mechanisms underlying mood disorders but also present a potential molecular target for developing new therapeutic strategies.
Professor Shu You-Sheng from the Institute for Translational Brain Research, Fudan University, is the corresponding author of the paper. Drs Li Junlong, Xiao Yujie, and Lv Shuxuan from the research group are the co-first authors. Wang Kun, Zhang Heyuan, Zheng Yuxiang, Zhang Xiaoxue, and Yang Hongkun made significant contributions to the work. This research was supported by grants from the National Natural Science Foundation of China, the Science and Technology Innovation 2030 -“ Major Project, the National Key Research and Development Program of China, Changping Laboratory, the Shanghai Outstanding Academic Leaders Program, and the China Postdoctoral Science Foundation.
Link to the original article:
https://rdcu.be/e2j6c or https://www.nature.com/articles/s41380-026-03453-2