Trigeminal neuralgia (TN) is a debilitating chronic pain disorder characterized by sudden episodes of severe facial pain, often triggered by innocuous stimuli such as light touch or routine daily activities like brushing teeth. Despite its clinical severity, the neural mechanisms underlying TN remain incompletely understood.
Recently, a research team led by Dr. Juan Deng and Dr. Liting Sun from the Institute of Translational Brain Research, Fudan University, published a study in Advanced Science identifying a critical brainstem circuit and molecular driver involved in the pathogenesis of TN.

Uncovering the Neural Circuit of Craniofacial Pain
Current pharmacological treatments for TN, including non-selective cation channel blockers such as carbamazepine and oxcarbazepine, can provide partial symptom relief. However, a substantial proportion of patients continue to experience moderate-to-severe pain even after long-term treatment, often accompanied by adverse effects such as dizziness, fatigue, and hepatotoxicity. These limitations highlight the need for a deeper understanding of the neural mechanisms underlying TN.
While somatic pain pathways have been extensively studied, the neural circuits responsible for craniofacial pain processing remain less well characterized. Peripheral facial sensory information is transmitted to the central nervous system via the trigeminal ganglion (TG), but the central circuits responsible for chronic craniofacial pain disorders, including TN, are still poorly defined.
To address this question, the researchers focused on the brainstem, the primary relay center for craniofacial sensory information.
Identification of the Sp5C–PBN Pain Transmission Pathway
Using a mouse model of TN induced by chronic constriction injury of the infraorbital nerve (CION), the team combined multiple advanced approaches-including single-cell transcriptomics, optogenetics, chemogenetics, and viral circuit tracing-to dissect the relevant neural circuitry.
To facilitate precise behavioral assessment, the researchers also developed a specialized head-fixation device for monitoring facial behaviors and refined the mouse grimace scale for more accurate quantification of spontaneous facial pain.
Their analyses highlighted two key brainstem regions:
Spinal trigeminal nucleus caudalis (Sp5C) — the primary relay center for craniofacial nociceptive signals.
Parabrachial nucleus (PBN) — a critical hub involved in pain processing and affective responses.
The study demonstrated that neurons in the superficial layers of the Sp5C project directly to the ipsilateral PBN. Functional manipulation of this pathway revealed that optogenetic inhibition significantly reduced facial pain behaviors, whereas activation induced pronounced pain-like responses. These findings establish the Sp5C–PBN pathway as a key transmission route for craniofacial pain signals.
Tac1 as a Critical Molecular Driver
To further characterize the molecular identity of Sp5C projection neurons, the team performed high-resolution single-cell RNA sequencing (Smart-Seq). Among the genes enriched in these neurons, Tac1 emerged as the most prominently expressed candidate.
Subsequent experiments confirmed that Tac1-expressing neurons in the Sp5C project to Tac1-positive neurons in the PBN, forming excitatory monosynaptic connections. Functional experiments demonstrated that:
CRISPR-Cas9–mediated knockdown of Tac1 significantly reduced pain hypersensitivity in TN mice; Chemogenetic inhibition of the Sp5CTac1–PBN pathway alleviated facial pain; Optogenetic or chemogenetic activation of the Sp5CTac1–PBN or TG–Sp5CTac1 circuit induced robust pain-like behaviors.
Together, these results identify the TG–Sp5CTac1–PBN Tac1 neural pathway as a critical mechanism underlying trigeminal neuralgia.
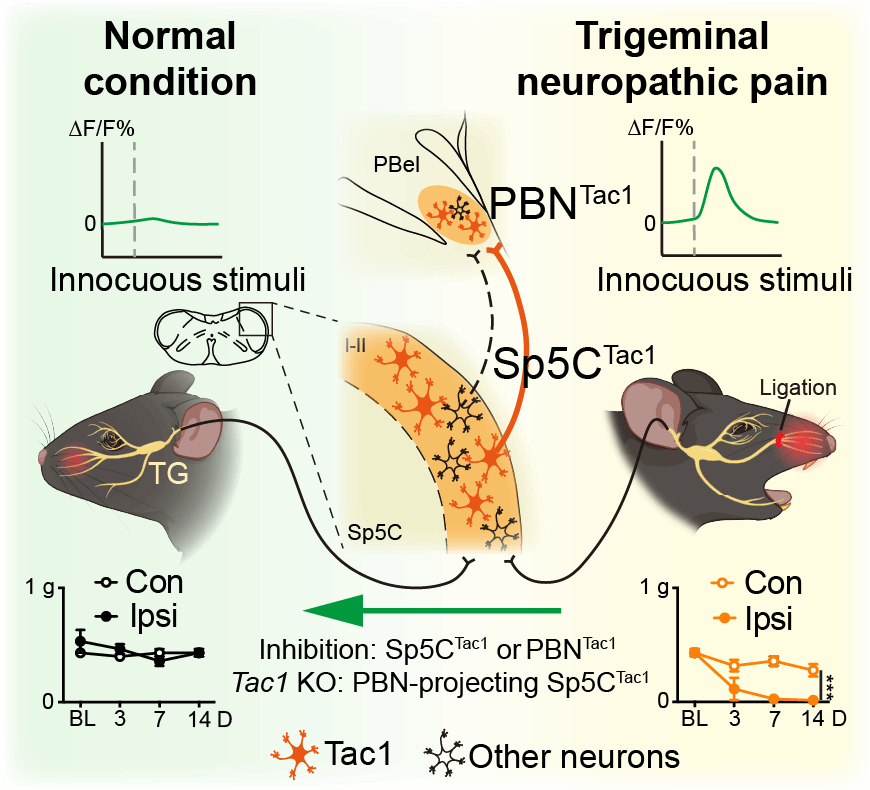
Figure 1 Mechanism of the TG-Sp5C-PBN circuit in driving TN pain
Scientific Significance
This study provides a comprehensive mapping of a previously unrecognized brainstem circuit involved in craniofacial pain. By integrating circuit-level and molecular analyses, the researchers reveal Tac1-expressing neurons as a key driver of trigeminal neuralgia.
These findings not only advance our understanding of the neural mechanisms underlying TN but also offer potential targets for the development of more precise therapeutic strategies for craniofacial pain disorders.
Publication Information
The study, titled “A Tac1-Expressing Brainstem Pathway Underlies the Pathogenesis of Trigeminal Neuralgia”, has been published in Advanced Science.
DOI: https://doi.org/10.1002/advs.202516310
Authors
Co-first authors: Liting Sun, Jiajia Wang, and Xiangyu Li (Institute of Brain Science, Fudan University).
Other contributors: Xinyi Lin, Juan Li, Qiutong Yu, and Zihan Wang.
Corresponding authors: Juan Deng and Liting Sun.
Grants
This project is supported by the National Science and Technology Innovation 2030 Major Program (No. 2022ZD0207300) and the National Natural Science Foundation of China (Nos. 31825013, 82101320, and 82222020).