Central nervous system (CNS) injuries, including traumatic brain injury, spinal cord injury and ischemic brain injury, are major causes of long-term neurological dysfunction. Unlike the peripheral nervous system, the adult CNS has a limited capacity for regeneration: injured axons rarely regrow efficiently, synaptic connections are difficult to re-establish, and damaged neural circuits often remain disconnected. Thus, the key to CNS repair is not only to "keep neurons alive", but also to reactivate their regenerative capacity and guide them to rebuild functional neural circuits.
On May 4, 2026, Professor Wei-Guang Li's team collaborated with Professor Jun Chen's team from the School of Pharmacy, Fudan University, Chief Physician Qiyong Mei's team from the Department of Neurosurgery, The Second Affiliated Hospital of Naval Medical University (Shanghai Changzheng Hospital), Professor Xiaoling Gao's team from Shanghai Jiao Tong University College of Basic Medical Sciences, and Chief Physician Zhihua Wang's team from Shanghai Pudong Hospital, Fudan University, to publish a research article in Nature Communications entitled "Self-assembling scaffolds epigenetically reactivate and electroactively guide neuronal regeneration to restore central neural circuits". The study reports a multifunctional, intravenously injectable nanosystem (MIIN) that can self-assemble in situ into large-pore neural guidance scaffolds at sites of CNS injury. Through epigenetic reactivation, remodeling of the lesion microenvironment, and activity-dependent synaptic reconnection, MIIN effectively promotes neural circuit repair after traumatic brain injury. The system encapsulates the class IIa HDAC4/5-selective inhibitor LMK-235 and features an electroactive polyaniline (PANI) interface together with asymmetrically distributed 5-hydroxytryptamine (5-HT) moieties. Upon reaching injured tissue, MIIN assembles into large-pore scaffolds that inhibit nuclear-retained class IIa HDAC activity, regulate glial activation, and provide an electroactive interface compatible with activity-dependent synaptic reconnection.

The central concept of the study is to advance "drug delivery" into in situ construction of a regenerative microenvironment. Conventional intracerebral scaffold implantation is limited by surgical trauma, infection risk, tissue compression and potential increases in intracranial pressure, making it difficult to apply broadly in brain repair. In contrast, MIIN is delivered intravenously and exploits pathological features of severe brain injury—namely blood-brain barrier disruption and elevated local myeloperoxidase (MPO)—to assemble spontaneously into scaffolds within the injured region. Importantly, the Janus asymmetric 5-HT modification prevents nanoparticles from simply forming dense aggregates; instead, they assemble into porous channels averaging approximately 3.5 μm, providing a "regenerative bridge" for axons to traverse the lesion site.
Mechanistically, the study focuses on the epigenetic barrier underlying regeneration failure in adult CNS neurons. After injury, class IIa HDACs—especially pathways associated with HDAC5—can continue to restrict histone H3 acetylation and regeneration-associated gene expression, making it difficult for neurons to initiate axonal regrowth. After MIIN forms scaffolds in the lesion, it continuously releases the HDAC4/5 inhibitor LMK-235, increases histone H3 acetylation in injured tissue, and upregulates regeneration-associated transcription factors such as Jun, Fos and Klf6. At the same time, MIIN reduces excessive activation of astrocytes and microglia/macrophages, improves the inflammatory microenvironment, and creates more favorable conditions for axonal extension and synaptic reconnection.
The results show that, in a mouse controlled cortical impact (CCI) model of traumatic brain injury, MIIN accumulates in injured brain tissue and forms scaffolds in situ. These scaffolds persist within the lesion, promote infiltration of SMI-312-positive axons, and increase retention of NeuN-positive mature neurons. Behavioral tests further demonstrate that MIIN improves performance in Morris water maze, forelimb placing ability and neurological scores, indicating that the treatment produces long-term neurological recovery rather than simple tissue protection. Whole-cell patch-clamp recordings show that MIIN restores both excitatory and inhibitory synaptic activity after injury. In addition, suppressing neuronal excitability in the secondary motor cortex (M2) using Kir2.1 weakens MIIN-mediated functional recovery, indicating that this repair process depends on neuronal activity. Using DREADD-based chemogenetics, the team further demonstrates that MIIN promotes functional recovery of the projection from the M2 cortex to the dorsal striatum/caudate-putamen (CPu), and that activity in this pathway is an important basis for improved motor function.
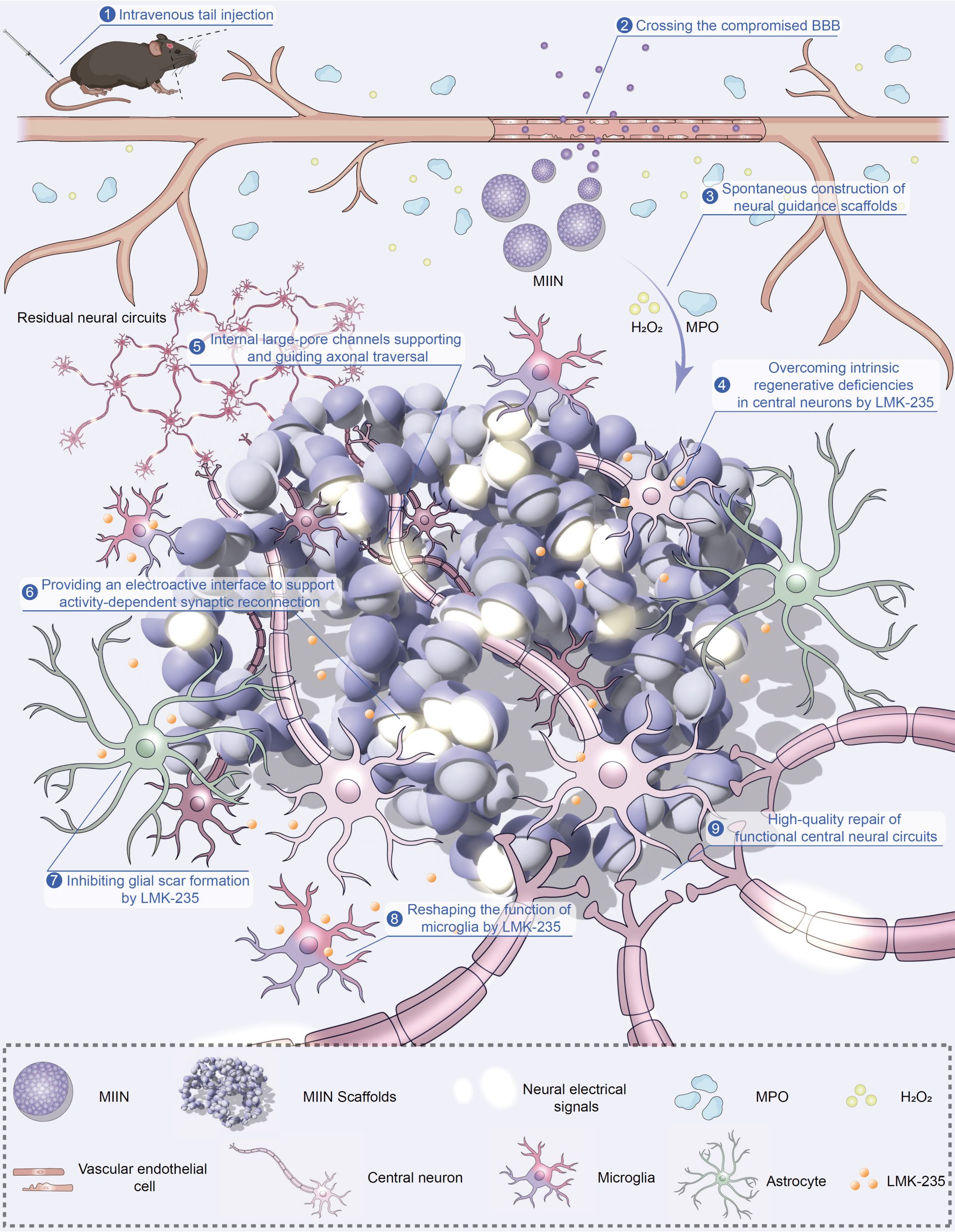
Figure 1. Overall process by which MIIN is administered intravenously, crosses the compromised blood–brain barrier, responds to MPO to form scaffolds, continuously releases LMK-235, regulates glial cells, and promotes neural circuit repair.
The significance of this study lies in shifting CNS repair from single-purpose "anti-inflammatory" or "neuroprotective" intervention toward functional neural circuit reconstruction. MIIN addresses three central challenges in CNS repair: first, LMK-235 relieves epigenetic suppression of neuronal regeneration; second, self-assembled large-pore scaffolds provide structural guidance for axonal growth; third, the electroactive interface and neuronal activity-dependent mechanisms support restoration of synaptic function. This strategy offers a new materials-design concept for neural circuit repair after traumatic brain injury and may also inspire future approaches for spinal cord injury, ischemic brain injury and neurological diseases involving chronic inflammation and circuit degeneration.
Dr. Shiqiang Tong, a graduate of the School of Pharmacy, Fudan University, and Dr. Shuai Ye, a postdoctoral fellow jointly trained by our institute and Jinshan Hospital, Fudan University, are co-first authors of the paper. Professor Wei-Guang Li of our institute, together with Professor Jun Chen from the School of Pharmacy, Fudan University, Chief Physician Qiyong Mei from The Second Affiliated Hospital of Naval Medical University, Professor Xiaoling Gao from Shanghai Jiao Tong University School of Medicine, and Chief Physician Zhihua Wang from Shanghai Pudong Hospital, Fudan University, are co-corresponding authors. This work was supported by the National Natural Science Foundation of China, the Shanghai Academic/Technology Research Leader Program, the Shanghai Science and Technology Commission, the Fudan University Medical Engineering Fund and other funding sources. The study also received guidance and support from Professor Bing Li of Research Center for Clinical Medicine, Jinshan Hospital Affiliated to Fudan University, Professor Tian-Le Xu of Shanghai Jiao Tong University School of Medicine and Professor Michael Xi Zhu of Shenzhen Bay Laboratory.
Article link: https://doi.org/10.1038/s41467-026-72397-6