Neurons typically receive synaptic inputs through their dendrites and soma, where these signals are integrated before triggering action potentials (APs) at the axon initial segment (AIS). The AIS serves as the primary site of AP initiation because it contains an exceptionally high density of voltage-gated Na⁺ channels, including low-threshold channel subtypes critical for spike generation (Hu et al., Nature Neuroscience, 2009). Although axons are generally not considered major sites of synaptic input, certain neurons receive inhibitory GABAergic synapses directly onto the AIS. These synapses regulate neuronal firing, contribute to arousal-state control, and are implicated in neurological and psychiatric disorders such as epilepsy, autism spectrum disorder, and schizophrenia.
Whether the AIS also receives excitatory glutamatergic inputs, however, has remained unresolved. Previous studies have described spine-like protrusions on the AIS of neurons across multiple species—including rodents, cats, non-human primates, and humans—as well as in brain regions such as the cortex and hippocampus. Because of their resemblance to dendritic spines, these structures were termed axonic spines. Yet their ultrastructure, physiological properties, and functional significance within neural circuits were largely unknown.
On May 15, 2026, the laboratory of Yousheng Shu at Fudan University published a study in Nature Neuroscience entitled “Excitatory synapses onto axonic spines jump-start action potentials and route information flow.” The study demonstrates that more than half of inhibitory neurons in the lateral septum (LS)—a brain region involved in emotional regulation and social behaviors—contain axonic spines, and systematically reveals their structural and functional roles at the synaptic, cellular, and circuit levels. The work will also be featured as the cover article of the journal’s June issue.

Using whole-cell patch-clamp recordings combined with fluorescence imaging, the researchers found that approximately 60% of LS neurons possess spine-like structures on their AIS. Neurons containing these structures were termed axonic-spine neurons (ASNs), whereas neurons lacking them were classified as non-ASNs. Similar proportions of axonic-spine-bearing inhibitory neurons were also observed in other GABAergic brain regions, including the bed nucleus of the stria terminalis (BNST) and the striatum, suggesting that axonic spines may be a widespread feature of inhibitory neurons throughout the central nervous system.
At the synaptic level, scanning electron microscopy reconstructions revealed that axonic spines form classic asymmetric excitatory synapses. Presynaptic terminals contained synaptic vesicles and active zones, while the postsynaptic membrane exhibited prominent postsynaptic densities (PSDs), forming classic asymmetric excitatory synapses. Consistent with these ultrastructural findings, two-photon glutamate uncaging experiments showed that stimulation of axonic spines evoked excitatory postsynaptic potentials (EPSPs) mediated by AMPA, kainate, and NMDA receptors. Moreover, spike-timing-dependent plasticity (STDP) paradigms induced long-lasting structural changes in axonic spines, including shortening of the spine neck, indicating that these structures are capable of activity-dependent remodeling.
At the cellular level, fluorescence and electron microscopy confirmed that axonic spines are specifically localized to the AIS, identified by Ankyrin-G labeling. Because the AIS is the site where APs are most readily initiated, the authors hypothesized that excitatory inputs onto axonic spines might drive neuronal firing more efficiently than inputs onto dendritic spines. Single-photon glutamate uncaging experiments supported this idea. Under weak stimulation, EPSPs generated at axonic spines were substantially larger than those evoked at dendritic spines. At intermediate stimulation intensities, activation of axonic spines alone was sufficient to trigger APs, whereas dendritic spine stimulation remained subthreshold. Even when both could evoke firing under stronger stimulation, APs triggered by axonic spines occurred earlier. These findings indicate that axonic spines enable neurons to fire faster and at lower levels of excitatory input than dendritic spines. Pharmacological experiments further suggested that this advantage depends on the dense concentration of voltage-gated Na⁺ channels at the AIS.
To investigate the circuit function of axonic spines, the researchers used mGRASP-based tracing to identify their upstream inputs. They found that projections from the dorsal CA3 (dCA3) region of the hippocampus formed contacts with Ankyrin-G-positive AIS structures in LS neurons, identifying dCA3 as a major upstream source of axonic spine input. dCA3 projections also innervated the soma and dendrites of LS neurons. Based on the enhanced spike-triggering capability of axonic spines, the team proposed a local circuit model in which dCA3 inputs preferentially activate ASNs, allowing them to fire earlier than neighboring non-ASNs. These early-firing ASNs then suppress surrounding neurons through feedforward inhibition, thereby biasing information flow within the local circuit—a process conceptually analogous to neural “routing.” Optogenetic experiments supported this model. Activation of dCA3-to-LS projections preferentially induced firing in ASNs. Moreover, even after inhibitory synaptic transmission was blocked pharmacologically, ASNs still fired significantly earlier than non-ASNs, suggesting that their rapid recruitment drives lateral inhibition that suppresses neighboring neurons. As a result, information carried by ASNs is preferentially transmitted to downstream circuits.
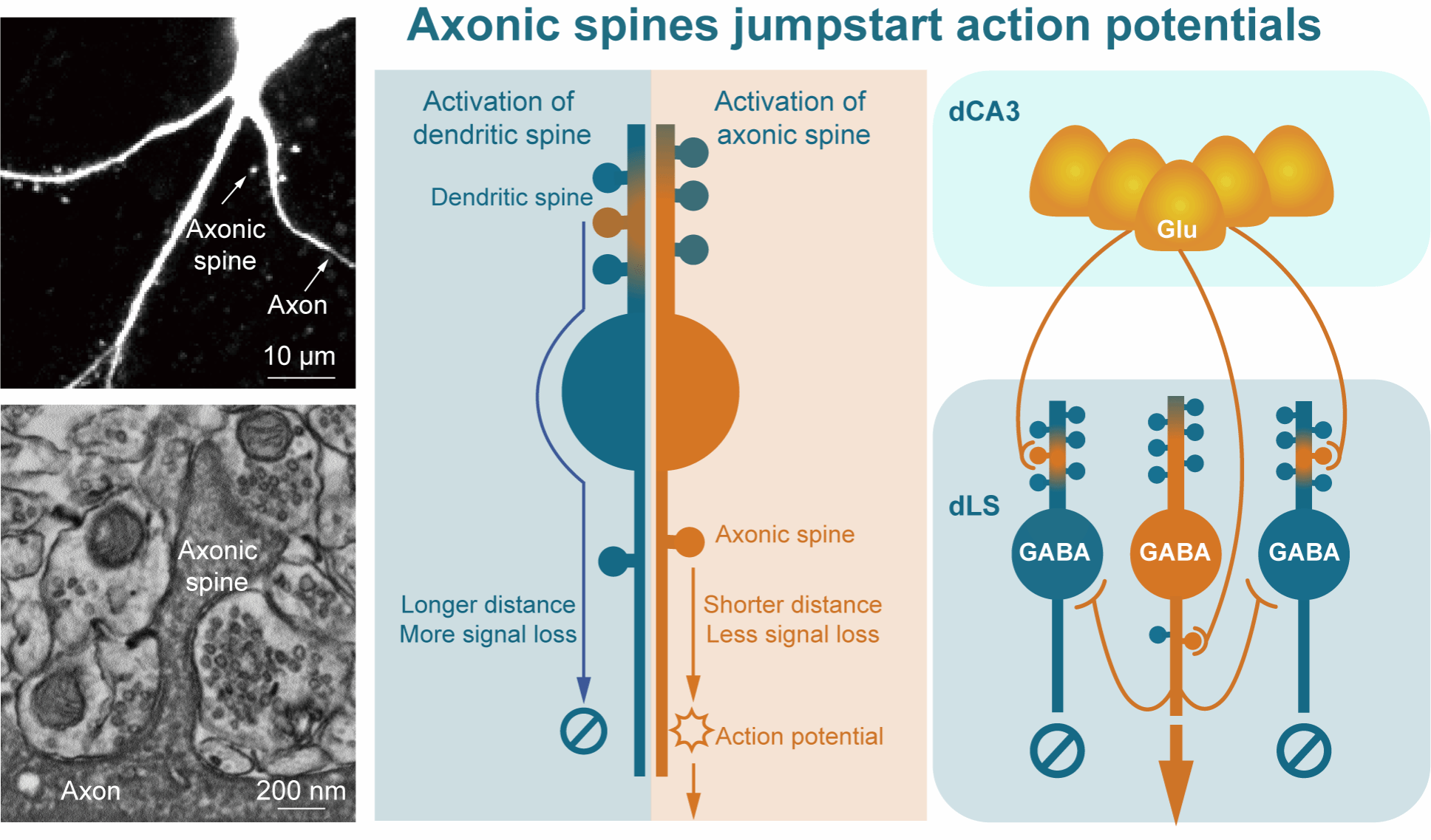
Figure 1 | Structural and functional model of axonic spines. Left: two-photon and electron microscopy images of axonic spines. Middle: cellular function of axonic spines. Compared with EPSPs generated at dendritic spines, which propagate through dendrites and soma and therefore undergo substantial attenuation over long distances (blue arrows), EPSPs generated at axonic spines can directly activate Na⁺ channels at the AIS to trigger APs, with minimal signal attenuation due to the much shorter propagation distance (yellow arrows). Right: circuit function of axonic spines. Glutamatergic dCA3 axons project onto both axonic and dendritic spines of GABAergic neurons in the dorsal lateral septum (dLS), but preferentially activate ASNs first. ASNs then rapidly inhibit neighboring non-ASNs, thereby mediating information routing within the local circuit.
Together, these findings provide the first direct evidence that axonic spines form functional excitatory synapses and reveal that they can trigger action potentials more rapidly and efficiently than dendritic spines. By enabling preferential information transmission within local circuits, axonic spines introduce a previously unrecognized mechanism for neural computation at the AIS. The study substantially expands current understanding of axonal function and offers a new conceptual framework for how neuronal output and information routing are regulated in the brain. Besides, the authors suggest that the computational principles of axonic spines may also inspire future designs for neuromorphic computing and artificial intelligence hardware.
Professor Yousheng Shu from the Institute for Translational Brain Research at Fudan University is the corresponding author of this study. Dr. Hongkun Yang, a postdoctoral researcher in the laboratory, is the first author of the paper. Important contributions to the study were made by doctoral student Kun Wang, Associate Researcher Wei Ke, Dr. Liang Li, and Dr. Shuyang Wang from Yousheng Shu’s laboratory, as well as by Yijie Chen from the School of Life Sciences at Nanjing University.The authors also acknowledge substantial support from Researcher Yilin Tai and Researcher Miao He at the Institutes of Brain Science, Fudan University; Researcher Yong Zhang from the Department of Neurobiology, School of Basic Medical Sciences, Peking University; Researcher Chao Tan, Researcher Bo Li, and doctoral student Wenxin Zhang from the Institute for Translational Brain Research, Fudan University; Professor Jian Wang and Professor Jintai Yu from the Department of Neurology, Huashan Hospital, Fudan University; and Dr. Yu Kong and Dr. Xu Wang from the Center for Excellence in Brain Science and Intelligence Technology, Chinese Academy of Sciences. This work was supported by the Science and Technology Innovation 2030 Major Program, the National Key R&D Program of China, the National Natural Science Foundation of China, and the Shanghai Academic/Technology Research Leader Program.