How cells sense and respond to changes in nutrient status to precisely regulate energy metabolism is a central question in the field of metabolic biology. Branched-chain amino acids (BCAAs), including leucine, isoleucine, and valine, are closely associated with metabolic homeostasis, and their roles in adipose tissue have garnered increasing attention in recent years. The first two steps of BCAA catabolism share the common enzymes BCAT (branched-chain amino acid transaminase) and BCKDH (branched-chain α-keto acid dehydrogenase complex). However, studies have shown that different metabolic enzymes exert distinct roles and mechanisms in adipose tissue, suggesting that the specific metabolic pathways of each BCAA may constitute key regulatory nodes1,2. Brown adipose tissue (BAT) mediates non-shivering thermogenesis through uncoupling protein 1 (UCP1) and is a critical organ for maintaining body temperature and energy balance. Activating BAT or inducing "browning" of white adipose tissue represents a potential strategy to combat obesity and metabolic disorders. Human and mouse studies have shown that the uptake and catabolism of BCAAs, particularly leucine, in BAT are significantly increased upon cold exposure, yet the specific roles of their catabolic enzymes in thermogenesis remain unclear 3.
The team of Guo Feifan and Xiao Fei at Fudan University has long been dedicated to studying the mechanisms by which amino acids regulate glucose, lipid, and energy metabolism4,5. On April 10, 2026, the team published a research paper entitled "Leucine catabolic enzyme AUH regulates BAT thermogenesis via PPARγ HMGylation and RNA-binding function in male mice" in Nature Communications. This study reveals for the first time that the leucine catabolic enzyme AUH (AU RNA binding methylglutaconyl-CoA hydratase) uses its metabolite 3-hydroxy-3- methylglutaryl coenzyme A (HMG-CoA) to perform a novel post-translational modification—HMGylation—on PPARγ, a core transcription factor for adipose thermogenesis, thereby enhancing its transcriptional activity. Simultaneously, AUH utilizes its RNA-binding function to directly stabilize Ucp1 mRNA. This dual mechanism synergistically promotes the expression of UCP1, a key effector of thermogenesis, thereby enhancing brown fat thermogenesis. This discovery not only reveals a novel link between leucine metabolism and the regulation of adipose thermogenesis but also identifies a new post-translational modification of PPARγ, offering a potential new target for the treatment of obesity and related metabolic diseases.

To elucidate the function of leucine catabolic enzymes in adipose thermogenesis, the research team first performed in vitro and in vivo genetic intervention experiments. They found that knocking down or knocking out AUH in mouse primary brown adipocytes or BAT significantly reduced UCP1 expression, cellular respiration rate, and the ability to maintain body temperature during cold exposure; conversely, AUH overexpression produced opposite effects. These results indicate that AUH is an important positive regulator of brown fat thermogenesis.
Further mechanistic studies revealed two parallel pathways by which AUH regulates UCP1 expression. On one hand, the AUH metabolite HMG-CoA directly HMGylates PPARγ at lysine 386, a highly evolutionarily conserved residue. This HMGylation significantly enhances the DNA-binding ability and transcriptional activity of PPARγ, thereby promoting Ucp1 transcription. On the other hand, AUH, through its RNA-binding function, directly binds to the AU-rich elements in the 3′ untranslated region (3′ UTR) of Ucp1 mRNA, significantly prolonging the half-life of Ucp1 mRNA and thus increasing UCP1 expression at the post-transcriptional level.
Interestingly, the team found that this regulatory axis is important under both physiological and pathological conditions. HMG-CoA levels in mouse BAT were significantly elevated upon cold exposure, whereas they were markedly reduced in the white adipose tissue of obese mice fed a high-fat diet. Overexpression of AUH in adipose tissue via adeno-associated virus effectively protected against high-fat diet-induced obesity, improved insulin sensitivity, and promoted white adipose browning. Moreover, analysis of transcriptomic data from human adipose tissue revealed that AUH gene expression was significantly lower in the white adipose tissue of obese individuals, suggesting a correlation between AUH expression and obesity.
This study reveals that HMG-CoA not only serves as a metabolic intermediate involved in energy production but also acts as a signaling molecule sensed by the cell to directly regulate the function of key transcription factors through covalent modification. This metabolite-driven post-translational modification, independent of classical regulatory mechanisms such as acetylation and phosphorylation, expands our understanding of metabolite signaling. Furthermore, the direct stabilization of Ucp1 mRNA by AUH as an RNA-binding protein highlights the importance of post-transcriptional regulation in adaptive thermogenesis. Targeting the AUH-HMGylation axis may provide a new intervention strategy for metabolic diseases such as obesity and insulin resistance.
Dr. Haizhou Jiang from the Institute for Translational Brain Research and Zhongshan Hospital, Fudan University, is the first author of this paper. Professors Guo Feifan and Xiao Fei, from the Institute for Translational Brain Research and Zhongshan Hospital, are the corresponding authors. This work was supported by grants from the National Natural Science Foundation of China, the National Key R&D Program of China, and the China Postdoctoral Science Foundation.
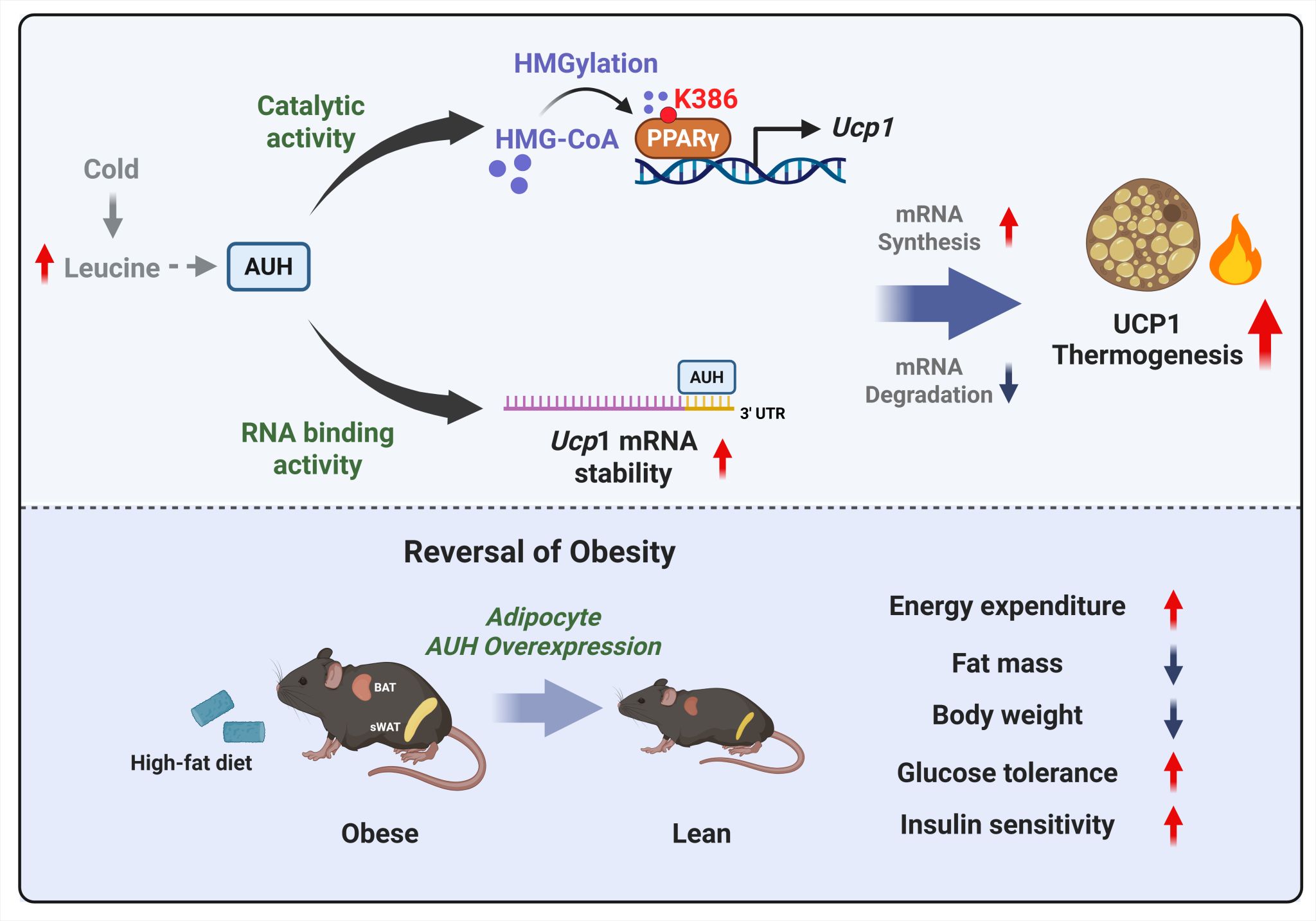
Figure 1: The leucine catabolic enzyme AUH regulates brown fat thermogenesis through dual mechanisms: HMGylation of PPARγ and RNA-binding function. In brown adipocytes, the leucine catabolic enzyme AUH promotes UCP1 expression and thermogenesis through two parallel pathways. First, AUH produces HMG-CoA, which directly HMGylates PPARγ at lysine 386 (K386), enhancing PPARγ transcriptional activity and thereby upregulating Ucp1 transcription. Second, AUH acts as an RNA-binding protein that directly binds to AU-rich elements (AREs) in the 3′ UTR of Ucp1 mRNA, stabilizing Ucp1 mRNA and increasing UCP1 expression at the post-transcriptional level. These two pathways synergistically increase UCP1 protein levels, driving brown fat thermogenesis and promoting white adipose browning, ultimately enhancing whole-body energy expenditure and protecting against high-fat diet-induced obesity.
References
1. Jiang H, Guo F, Xiao F. Role of Branched-Chain Amino Acid Catabolism in the Regulation of Adipocyte Metabolism. Endocrinology. May 19 2025;166(7)doi:10.1210/endocr/bqaf089
2. Ma QX, Zhu WY, Lu XC, et al. BCAA-BCKA axis regulates WAT browning through acetylation of PRDM16. Nature metabolism. Jan 2022;4(1):106-122. doi:10.1038/s42255-021-00520-6
3. Yoneshiro T, Wang Q, Tajima K, et al. BCAA catabolism in brown fat controls energy homeostasis through SLC25A44. Nature. Aug 2019;572(7771):614-619. doi:10.1038/s41586-019-1503-x
4. Xiao F, Guo F. Impacts of essential amino acids on energy balance. Molecular metabolism. Mar 2022;57:101393. doi:10.1016/j.molmet.2021.101393
5. Hu X, Guo F. Amino Acid Sensing in Metabolic Homeostasis and Health. Endocrine reviews. Jan 28 2021;42(1):56-76. doi:10.1210/endrev/bnaa026