Pain is an unpleasant sensory and emotional experience, often accompanied by the generation of negative affective states[1-3]. Both pain and its subsequent emotional states are profoundly influenced by the complex interplay of biological, psychological, and social factors[4]. Previous research has shown that social reward derived from positive affiliative interactions is associated with the release of neurochemicals such as dopamine and endogenous opioids, which can reduce pain sensitivity and enhance well-being[1,3,5]. Conversely, negative social experiences or social isolation can exacerbate pain and emotional reactions, heighten perceptual sensitivity, trigger maladaptive coping strategies, and increase the risk of developing chronic pain[6,7]. However, the intrinsic mechanisms by which social reward-related neural circuits directly modulate pain and negative emotional responses remain unclear. The medial amygdala (MeA) has been implicated in the regulation of social behaviour[8]. Our recent studies have further revealed a critical role for the MeA in social reward processing and the promotion of prosocial behaviour[9-11]. Despite extensive research linking the MeA to emotional and pain regulation[12,13], the specific neuronal subpopulations within the MeA and their downstream circuits governing emotional states and pain perception have remained largely unexplored. Therefore, this study investigated the role of activating GABAergic neurons in the MeA and their projection to the medial preoptic area (MPOA) in pain-related behaviours.

On 18th November 2025, Researcher Hu Rongfeng from our institute and collaborators published a research paper entitled "Alleviation of negative emotional and pain responses through amygdala-hypothalamic social circuit activation" in the internationally renowned academic journal PNAS. This work investigated a newly identified, non-canonical social reward circuit - the neural pathway projecting from the medial amygdala (MeA) to the medial preoptic area (MPOA) and explored whether direct activation of this circuit could alleviate pain-related emotional and behavioural responses. The results demonstrated that both optogenetic and chemogenetic activation of GABAergic neurons in the MeA significantly reduced freezing behaviour following electric shock, anxiety-like behaviours, and pain responses in the hot plate test in both male and female mice. Furthermore, optogenetic activation of the MeA-to-MPOA neural circuit produced similar ameliorative effects. This study deepens our understanding of the neural circuit mechanisms underlying pain modulation and underscores the urgent necessity of integrating social and psychological factors into therapeutic strategies aimed at improving human health and well-being.
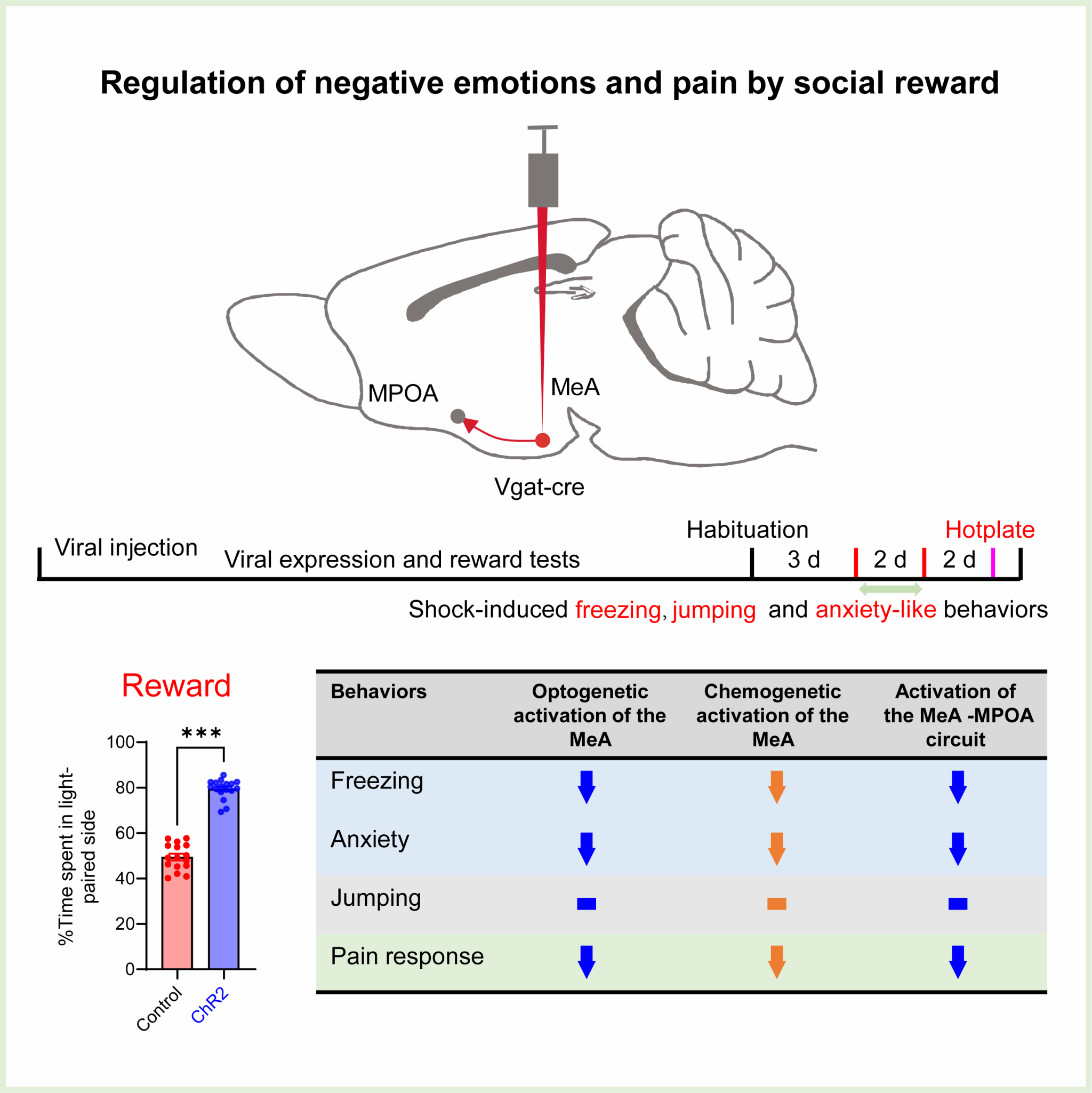
Figure 1. Graphical summary of this study.
Researcher Hu Rongfeng from Fudan University and Shanghai Pudong Hospital, and Young Researcher Hu Shuaishuai from the Shanghai Mental Health Centre, Shanghai Jiao Tong University School of Medicine, are the co-corresponding authors of this paper. Lu Xinqi ( PhD student) and Chen Guohua (PhD student) from the Institute for Translational Brain Research, Fudan University, are the co-first authors. Professor Weizhe Hong from the University of California, Los Angeles, also made significant contributions. Thanks also go to Researcher Deng Juan for providing technical support. This work was supported by grants from the National Natural Science Foundation of China (No. 82301395), among others.
Link to the original article
Xinqi Lu, Guohua Chen, Weizhe Hong, Shuaishuai Hu*, Rongfeng K. Hu*. (2025) Alleviation of negative emotional and pain responses through amygdala-hypothalamic social circuit activation. PNAS. 122 (47) e2511106122. https://doi.org/10.1073/pnas.2511106122
References:
1. L. L. Tan, R. Kuner, Neocortical circuits in pain and pain relief. Nat Rev Neurosci 22, 458-471 (2021).
2. C. Peirs, R. P. Seal, Neural circuits for pain: Recent advances and current views. Science 354, 578-584 (2016).
3. E. Navratilova, F. Porreca, Reward and motivation in pain and pain relief. Nat Neurosci 17, 1304-1312 (2014).
4. R. B. Fillingim, Individual differences in pain: understanding the mosaic that makes pain personal. Pain 158, S11-S18 (2017).
5. K. Onoda et al., Decreased ventral anterior cingulate cortex activity is associated with reduced social pain during emotional support. Soc Neurosci-Uk 4, 443-454 (2009).
6. E. L. Miller, Social Connectedness and Pain. Pain Manag Nurs 24, 111-112 (2023).
7. S. F. Allen, S. Gilbody, K. Atkin, C. van der Feltz-Cornelis, The associations between loneliness, social exclusion and pain in the general population: A N=502,528 cross-sectional UK Biobank study. J Psychiatr Res 130, 68-74 (2020).
8. N. Prakash, A. Abu Irqeba, J. G. Corbin, Development and function of the medial amygdala. Trends Neurosci 48, 22-32 (2025).
9. F. Sun, E. Y. Wu, W. Hong, A neural basis for prosocial behavior toward unresponsive individuals. Science 387 (2025).
10. Y. E. Wu et al., Neural control of affiliative touch in prosocial interaction. Nature 599, 262-267 (2021).
11. R. K. Hu et al., An amygdala-to-hypothalamus circuit for social reward. Nat Neurosci 24, 831-842 (2021).
12. D. Ivanova et al., Urocortin3 in the Posterodorsal Medial Amygdala Mediates Stress-induced Suppression of LH Pulsatility in Female Mice. Endocrinology 162 (2021).
13. M. Yoshida, Y. Takayanagi, T. Onaka, The Medial Amygdala-Medullary PrRP-Synthesizing Neuron Pathway Mediates Neuroendocrine Responses to Contextual Conditioned Fear in Male Rodents. Endocrinology 155, 2996-3004 (2014).