On 24th October 2025, a research group led by Ni Jinfei at the Institute for Translational Brain Research (ITBR), Fudan University, published a study online in Neuron entitled "An enteric-DRG pathway for interoception and visceral pain in mice". This research reveals a novel neuroanatomical and functional interaction mechanism between the enteric nervous system and the DRG sensory pathway.
Interoception is the physiological process by which the body perceives information from internal organs, relying primarily on two sensory pathways: the vagal sensory pathway and the spinal sensory pathway. These are mediated by sensory neurons located in the nodose ganglia, situated below the jugular foramen, and sensory neurons in the dorsal root ganglia (DRG), respectively.
As the classical interoceptive pathway, the vagal sensory nerve has been studied for decades. Previous research indicates that vagal sensory nerves can perceive sensory information from multiple organs, such as the mechanical tension of the digestive tract after eating, nutrients in food, aortic tension changes with each heartbeat, and blood oxygen/carbon dioxide concentrations. Over the past decade, researchers have employed novel neurobiological techniques to further elucidate the encoding mechanisms of these interoceptive signals by vagal sensory nerves and their mediated physiological functions. However, research progress on the other interoceptive pathway - "the spinal sensory pathway " has been relatively slower.
Both the spinal sensory neurons that perceive visceral sensations and those that perceive bodily surface sensations are located in the DRG. Historically, DRG research has primarily focused on their role in touch, temperature perception, and pain/itch, with comparatively little investigation into their function in interoception. This is partly because these sensory neurons are distributed across multiple ganglia, making them challenging to study, and partly because visceral organs contain not only DRG-derived sensory endings but also vagal sensory endings and sympathetic/parasympathetic endings. Furthermore, the gastrointestinal tract harbours an extensive intrinsic nervous system - the enteric nervous system (ENS). Therefore, studying the structure and function of DRG-derived sensory nerves requires several technical capabilities: first, a genetic method to specifically label DRG sensory nerve endings within visceral organs; second, the ability to perform functional imaging of DRG sensory neurons to investigate which visceral information activates them and their encoding patterns; and third, the capacity to specifically activate DRG sensory nerve endings located in viscera of living animals to study their physiological functions.
Before embarking on this research, although the group recognised the pathway's potential value, they had not previously ventured into DRG sensory neuron research. The first turning point came with the arrival of a team member who had studied DRG sensory neurons during their Master's (also the first author of this paper). At the time, the transgenic mice originally planned for the project were not yet fully available, so the team decided to attempt functional imaging of DRG sensory neurons to explore their ability to sense signals from the gastrointestinal tract and other viscera. Through this process, the team systematically mastered various experimental techniques, including DRG dissection, staining, and viral injections.
The second turning point arose from a transgenic mouse line. As a continuation of the group's previous research direction, they had originally planned to focus on the structure and function of the enteric nervous system. For this, they utilised a Phox2b-FlpO transgenic mouse line, which labels all peripheral nerves innervating the digestive tract except the DRG, including the ENS. The commonly used fluorescent reporter mice Ai9 and Ai32, which express fluorescent protein or ChR2 in Cre-positive cells, have their expression suppressed in the presence of FlpO (a detail uncovered after consulting the original JAX literature). Therefore, in Phox2b-FlpO mice, the only FlpO-negative peripheral nerves innervating the viscera are the DRG-derived sensory nerves. By combining this with Vglut2-Cre transgenic mice (which express Cre in all or specific subtypes of DRG sensory neurons), it became possible to specifically label or optogenetically activate DRG sensory nerves within viscera, thereby observing their distribution, terminal structures, and induced behavioural and physiological responses. This strategy is termed a "cross-genetics strategy".
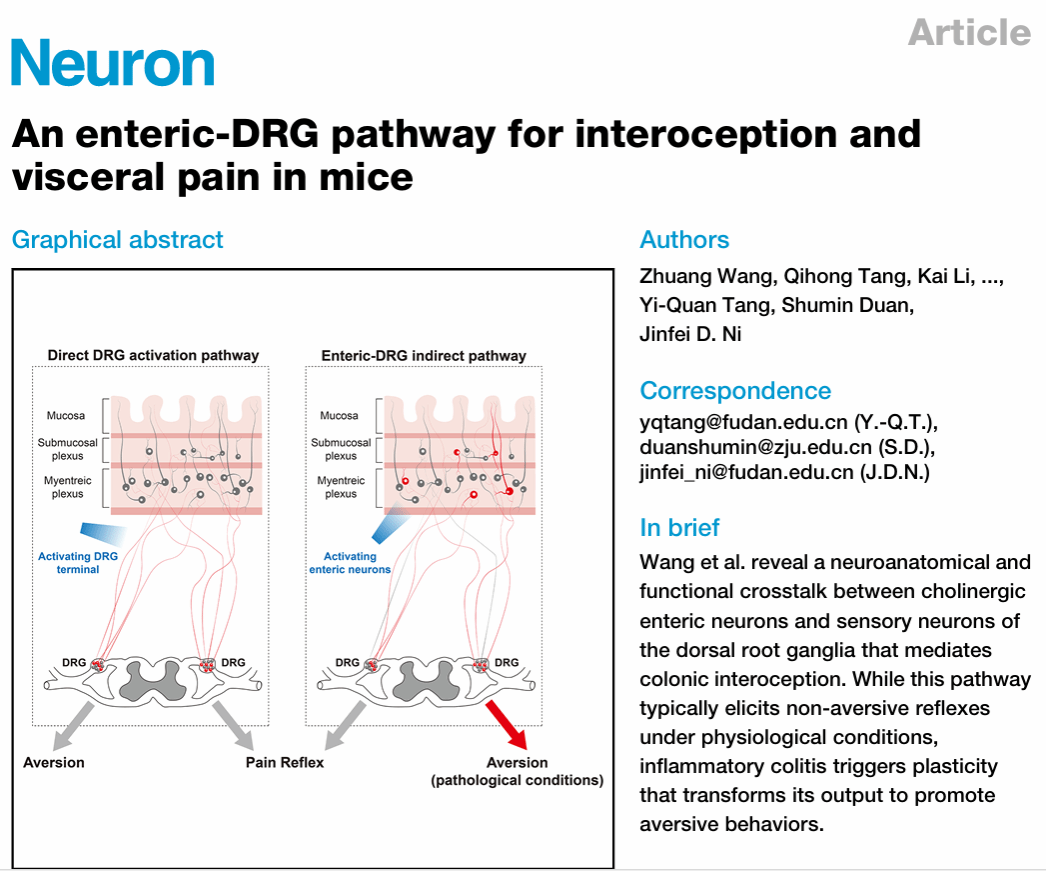
Using this strategy (Vglut2-cre; Phox2b-FlpO; Ai9/Ai32), the research team found that DRG sensory endings densely innervate the colorectum and stomach, with sparser distribution in the small intestine (duodenum, jejunum, and ileum). They also observed widespread projections of DRG sensory nerve endings to other visceral organs, including the trachea, oesophagus, heart, lungs, pancreas, spleen, kidneys, and bladder. Furthermore, using viral tracing techniques, the team confirmed specific terminal morphologies formed by DRG sensory neurons within the colorectum, such as intraganglionic varicose endings (IGVE), intramuscular arrays (IMA), perivascular endings, and mucosal endings. These projection analyses suggest that DRG sensory neurons play a significant role in interoceptive processes.
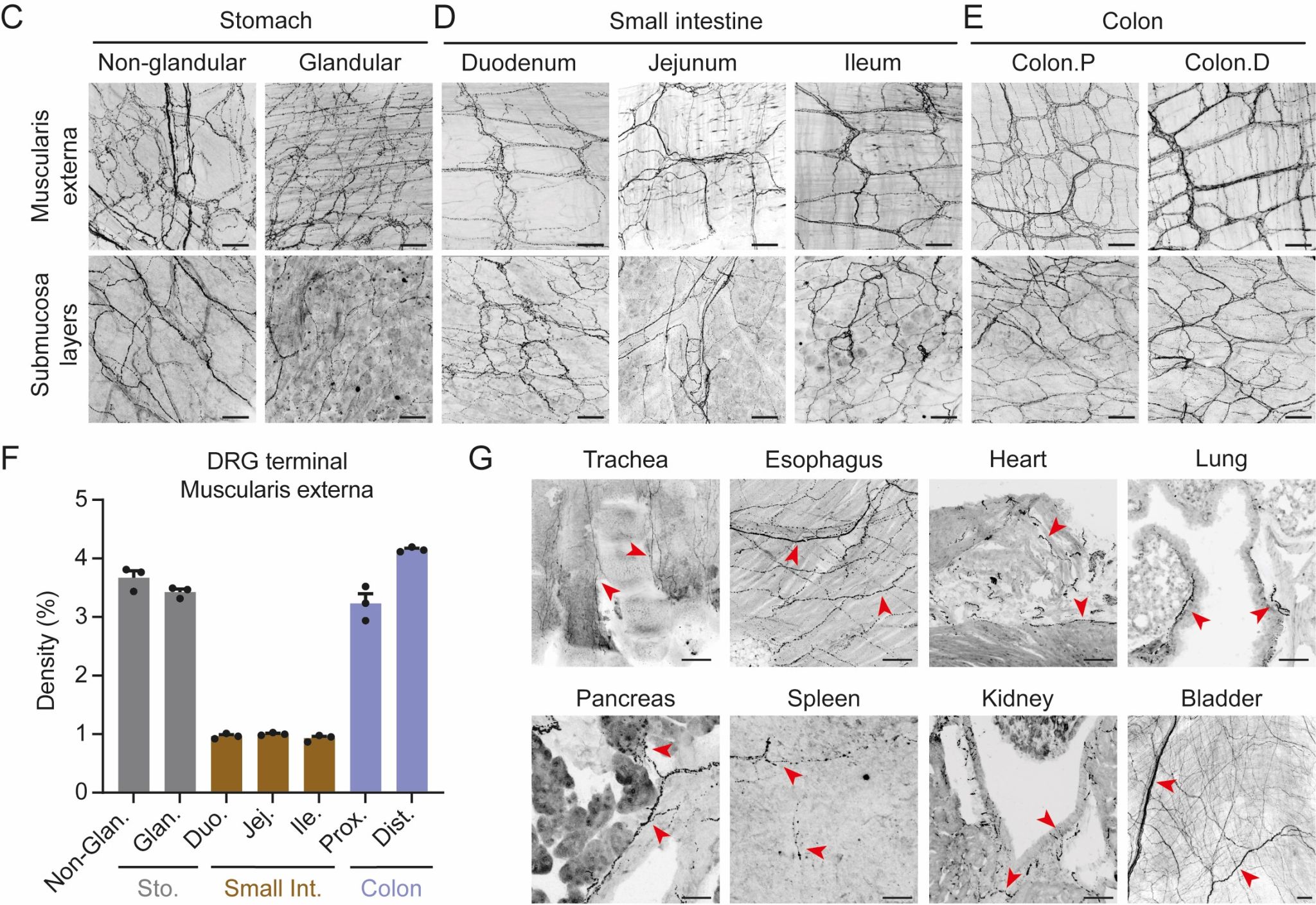
Figure 1: Projection terminals of DRG sensory neurons in various viscera
Subsequently, the team used optogenetics to activate DRG terminals innervating the rectum. The rectum was chosen not only for its high DRG innervation density but also because it allowed for fibre optic implantation via the anal opening to analyse behavioural responses in freely moving mice upon light activation. However, mice dislike having a fibre optic inserted into the anus and often try to remove it. To address this, the team devised an adaptive training protocol: for three days before the experiment, a fibre optic was inserted into the mouse's anus daily, and the tether was secured at the base of the tail. To prevent the mouse from removing the fibre, it was placed on a circular platform elevated about 30-40 cm high, only slightly wider than the mouse's body. If the mouse attempted to turn and remove the fibre, the constraints of the elevated ring discouraged it, and it gradually learned to walk around the platform dragging the fibre, thus becoming adapted.
Optogenetic activation of DRG sensory neurons innervating the colorectum elicited a characteristic "arching response" in mice - an upward arching of the back and lifting of the abdomen, a classic behavioural indicator of visceral pain. Concurrently, recording the electromyographic (EMG) activity of abdominal muscle contractions, indicative of the visceromotor response (VMR), also detected clear electrical signals, further confirming nociceptive activation.
Visceral pain is often accompanied by negative emotions such as aversion and distress. To investigate whether activating DRG sensory neurons induces aversion, the team employed a conditioned place avoidance assay: when a mouse moved from a circular platform to the ground, it received a light stimulus. The latency to step down from the platform (avoidance latency) was recorded. Results showed that control mice, regardless of the number of light stimulation trials, had an average latency under 30 seconds. In contrast, mice whose DRG sensory endings were optogenetically activated exhibited significant avoidance after just a single stimulation trial, indicating that this activation can serve as a "negative instructional signal" to trigger aversion.
Additionally, the team activated specific DRG sensory neuron subtypes, such as TrpV1-, Cysltr2-, and Piezo2-expressing neurons, and found that different subtypes induced varying degrees of behavioural response. However, only specific DRG sensory pathways were capable of triggering affective visceral pain.
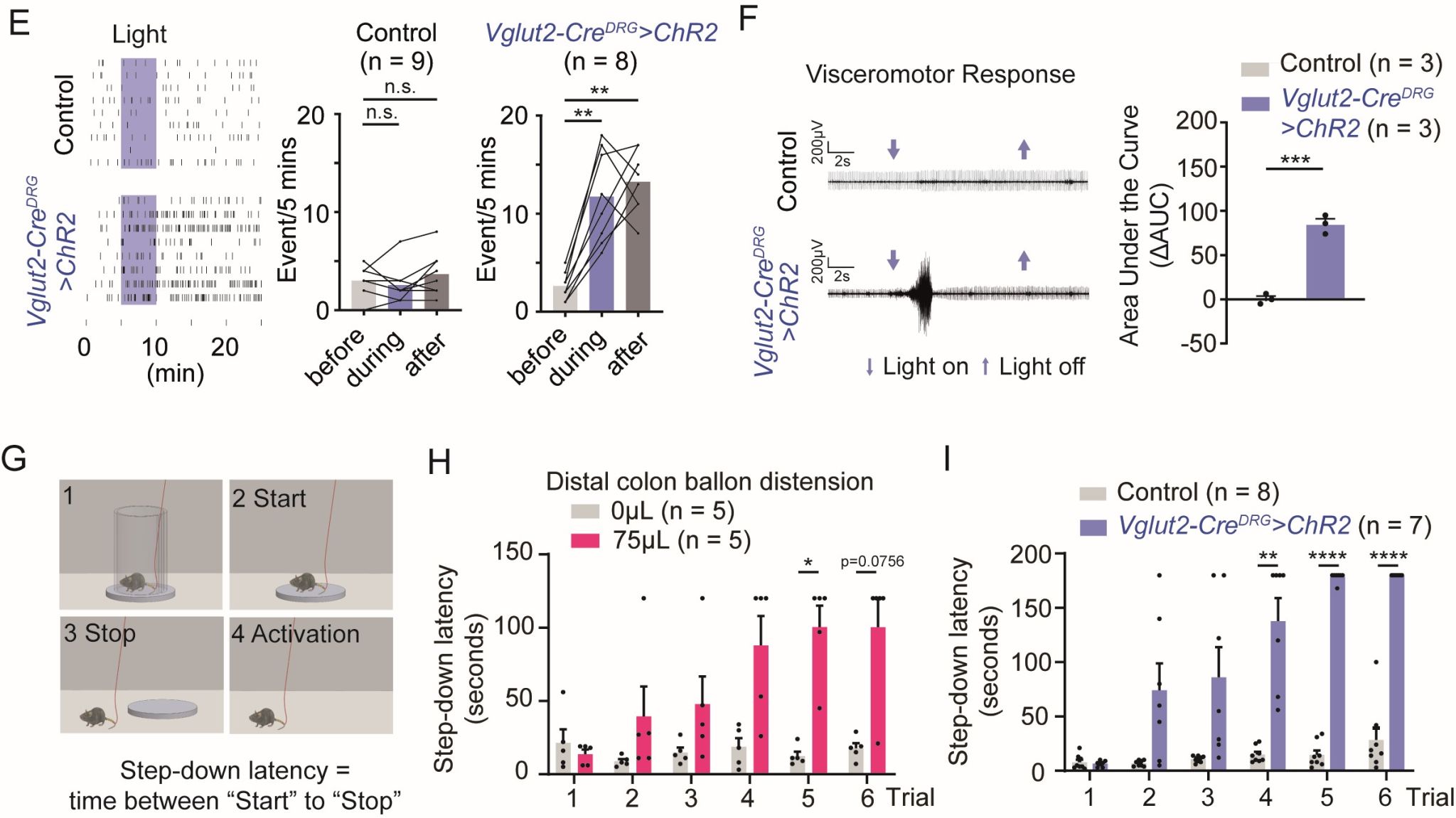
Figure 2: Optogenetic activation of DRG terminals innervating the rectum evokes arching reflex and negative affect
Despite the smooth progress in morphological and behavioural experiments, calcium imaging experiments repeatedly encountered setbacks. The team's procedure, from surgical exposure of the DRG to two-photon microscopy imaging, appeared flawless. However, balloon distension stimulation within the rectum invariably caused minute axial displacements in the tissue surrounding the DRG. As two-photon microscopy is exquisitely sensitive to axial movement, obtaining stable data proved impossible. The team then realised that a simple upright wide-field microscope equipped with a high-sensitivity camera might suffice, but they couldn't locate suitable equipment in the short term.
Just as calcium imaging hit this impasse, the team received two pieces of unwelcome news: laboratories led by renowned sensory neurobiologists David Ginty and Ardem Patapoutian in the United States had both published papers studying DRG sensory neurons innervating the rectum. Notably, the cross-genetics strategy used by David Ginty's team was identical to the one designed by this group, and their calcium imaging results were exquisitely presented, covering almost all aspects the Fudan team had originally planned to investigate. Although their behavioural analysis was somewhat cursory, the overall study was comprehensive and systematic. This development plunged the team into uncertainty about whether to continue the project, and the first author's morale notably dipped. While the team performed well in behavioural experiments, it seemed unlikely to yield a substantial breakthrough.
It was at this point that the third turning point brought a new direction. This suggestion came from Academician Duan Shumin during a group meeting. The team had previously observed that numerous DRG sensory endings in the gastrointestinal tract could envelop enteric neurons, forming characteristic IGVE structures, suggesting a potential functional connection between the enteric nervous system and DRG sensory neurons. However, this observation was not initially prioritised. Academician Duan repeatedly enquired during group meetings about the IGVE structure and the relationship between enteric nerves and DRG, but the team's initial responses were vague. During a group meeting following the publication of Ginty's paper, Academician Duan once again emphasised the importance of investigating the functional coupling between enteric nerves and DRG. With few other avenues remaining, the team heeded this advice seriously this time.
The team hypothesised that if a functional connection existed between enteric nerves and DRG, local injection of AAV-ChR2 to activate enteric neurons should induce similar behaviours. Experimental results showed that optogenetic activation of enteric neurons indeed evoked a clear arching behaviour, consistent with the effect of activating DRG terminals. Furthermore, if nociceptive neurons within the DRG were ablated using a toxin, activating enteric neurons no longer induced arching behaviour, demonstrating that this behaviour is mediated by DRG neurons. However, in the avoidance learning assay, activating enteric neurons, even after five light stimulation trials, failed to significantly prolong the mouse's latency on the platform. This indicated that while enteric neuron activation could elicit a nociceptive reflex behaviour, it was insufficient to trigger an aversive response. Further investigation revealed that specifically activating cholinergic enteric neurons induced the arching behaviour, and acetylcholine was identified as a key molecular mediator of this functional connection.
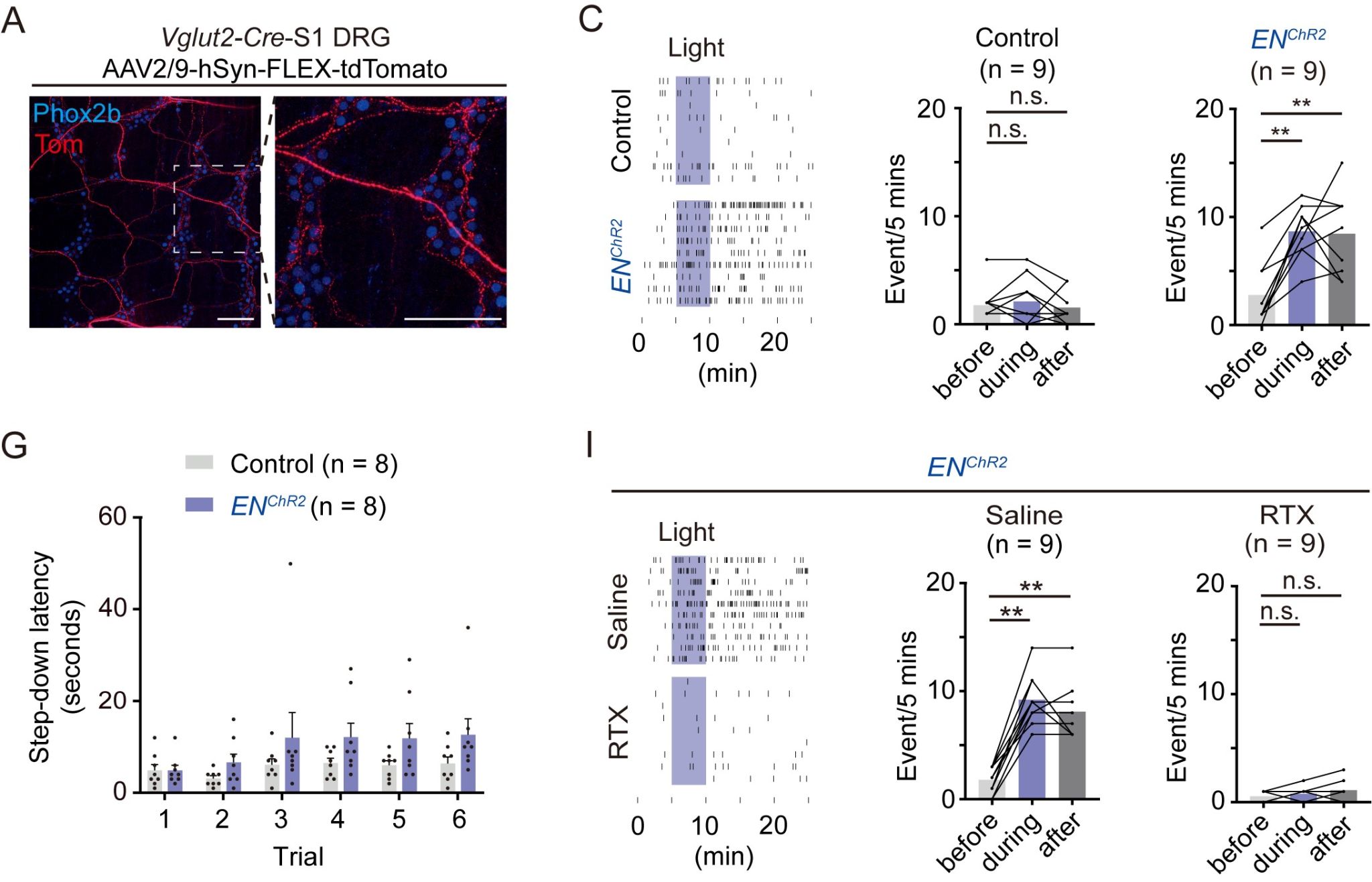
Figure 3: Activation of enteric neurons evokes arching reflex dependent on the DRG pathway
Previous studies have shown that colonic inflammation can induce hypersensitivity to colorectal sensory information in mice, potentially contributing to visceral pain sensitisation, but research has largely focused on DRG sensory neurons. The team discovered that although enteric neuron activation can evoke nociceptive reflex behaviours (such as arching and VMR) via the ENS-DRG signalling pathway, this pathway does not typically trigger aversive responses under physiological conditions. Therefore, the team proceeded to investigate whether colonic inflammation could sensitise this pathway, thereby inducing aversion.
Using a colitis mouse model (mice administered 3% DSS in drinking water for five consecutive days, followed by a return to normal water), the team conducted avoidance learning assays during the pre-induction, acute, and recovery phases. Results showed that during acute colitis, optogenetic activation of cholinergic enteric neurons significantly delayed the mouse's latency to step down from the platform, indicating that inflammation had rendered the signals mediated by the ENS-DRG pathway capable of eliciting aversion. More surprisingly, after a 21-day recovery period (by which time the mice had fully regained their body weight), activating this pathway still potently induced aversive behaviour. This suggested that the inflammation-induced changes in the ENS-DRG pathway persisted well beyond the acute phase. These results indicate that colitis can "hijack" a specific ENS-DRG signalling pathway, thereby triggering intense aversive behaviour.
In this study, the team experienced a complete journey: from serendipitous beginnings, to encountering competitive pressure, and finally achieving a breakthrough by shifting perspective. This underscores that in scientific research, maintaining cognitive flexibility and a keen eye for detail, especially during challenging times, can often lead to more innovative discoveries than strictly following a predetermined plan.
Researcher Ni Jinfei from the Institute forTranslational Brain Research, Fudan University, Academician Duan Shumin, and Researcher Tang Yiquan from the Institutes of Brain Science, Fudan University, are the co-corresponding authors of this paper. Doctoral student Wang Zhuang, Tang Qihong, and Dr Li Kai, all from the Institute of Translational Brain Research, Fudan University, are the co-first authors. This research also received advice and technical support from Professor Xu Guangyin of Soochow University, and Professor Lu Wei and Dr Sun Liting from the Institute of Translational Brain Research. The study was supported by funding from the 2021 Science and Technology Innovation 2030 "Brain Science and Brain-Inspired Research" Major Project, the National Natural Science Foundation of China, and the China Postdoctoral Science Foundation.
Dr Ni Jinfei's research group is dedicated to studying the autonomic nervous system, including brain-body interactions mediated by interoceptive systems, with a current focus on the structure and function of neural connections between digestive system organs and the brain. Scientists interested in these questions are welcome to discuss potential collaborations.

Link to the original article: https://www.sciencedirect.com/science/article/pii/S0896627325007482?dgcid=author