On 29th September 2025, a research group led by Yang Hanting at the Institute of Translational Brain Research, Fudan University, published a study in Nature Communications entitled "Molecular Mechanisms of SLC30A10-Mediated Manganese Transport". This study elucidated, at the molecular level, the mechanism by which SLC30A10 mediates manganese efflux and its critical role in mitigating manganese toxicity, providing an important theoretical foundation for understanding the pathogenesis of and exploring potential targeted interventions for the autosomal recessive disorder HMNDYT1 and other neurological diseases.

Manganese (Mn) is an essential trace element in the human body, involved in key processes such as protein synthesis, energy metabolism, oxidative stress defence, and nervous system development. However, an excess of Mn2+ can impair the function of organelles like the Golgi apparatus and endoplasmic reticulum, interfering with protein folding, post-translational modification, intracellular trafficking, and degradation. Concurrently, it can disrupt oxidative phosphorylation and increase the generation of reactive oxygen species (ROS), triggering mitochondrial failure and cell death. SLC30A10 (solute carrier family 30 member 10, also known as ZnT10) is a core transmembrane transporter responsible for maintaining cellular manganese homeostasis. Primarily distributed in the brain and liver, it specifically mediates the efflux of manganese ions, thereby reducing intracellular manganese levels. Mutations in the SLC30A10 gene cause the autosomal recessive disorder HMNDYT1 (hypermanganesemia with dystonia-1), characterised by Parkinson's-like movement and speech impairments, as well as liver damage in patients. Investigating the transport mechanism of SLC30A10 is not only crucial for elucidating its physiological function but also instrumental in identifying modulators targeting SLC30A10, potentially offering treatments for HMNDYT1 and related neurological conditions.
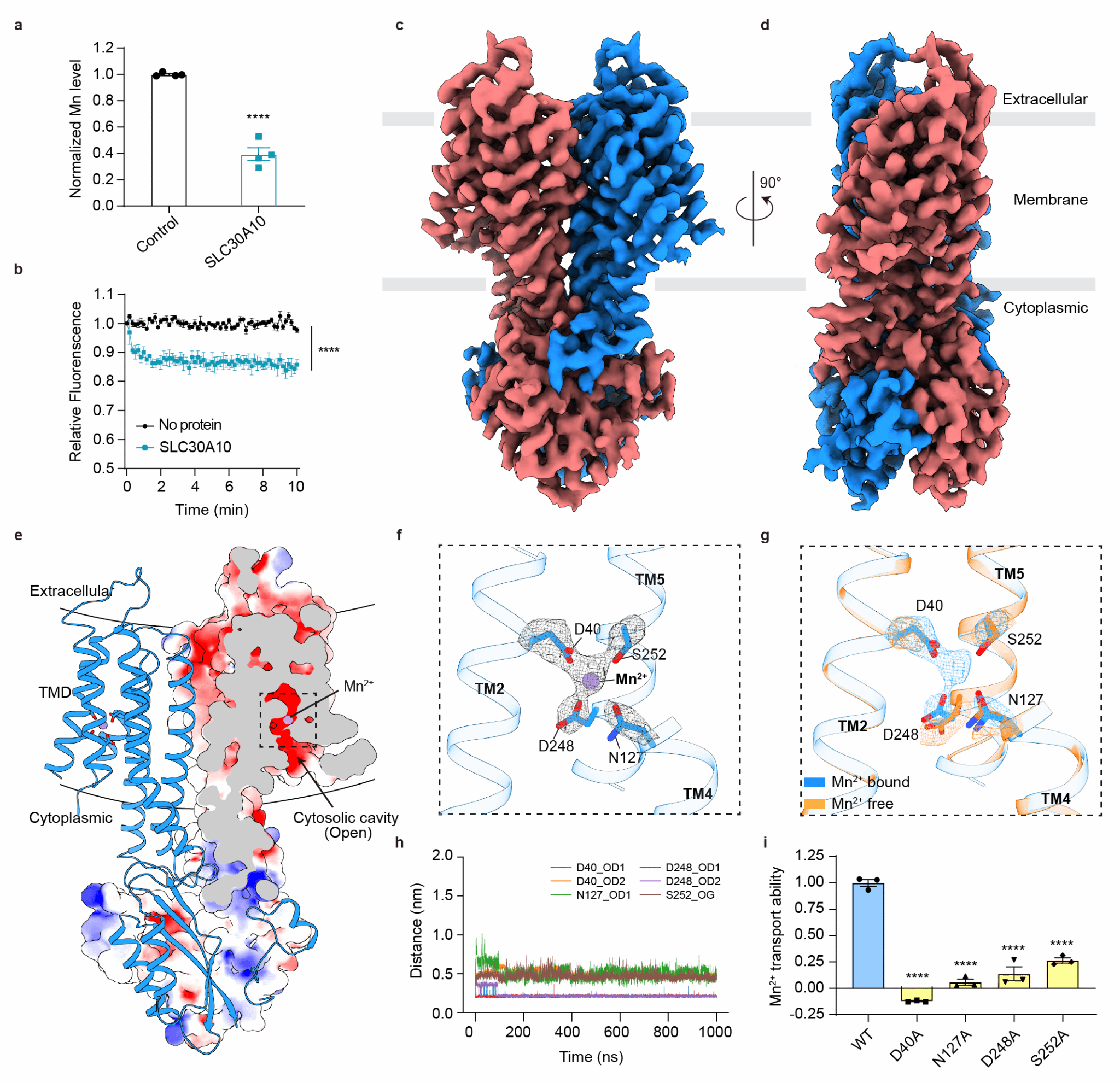
This study found that under conditions of Mn2+ overload, SLC30A10 mitigates cellular oxidative stress and maintains mitochondrial homeostasis and cell viability by mediating Mn2+ efflux. Ion transport assays conducted at both the cellular and protein levels confirmed the high selectivity of SLC30A10 for Mn2+ . To further understand the molecular mechanism of its ion transport, the research team employed cryo-electron microscopy (cryo-EM) to resolve the high-resolution structure of full-length human SLC30A10, revealing its architecture as a homodimer. Each monomer of SLC30A10 comprises a transmembrane domain (TMD) featuring six transmembrane helices and a cytoplasmic C-terminal domain (CTD). Notably, by comparing the structures obtained in the presence and absence of Mn2+, the team observed that Mn2+ forms an octahedral-like coordination with residues D40 (TM2), N127 (TM4), D248 (TM5), and S252 (TM5). This represents the first identification of a Mn2+ -specific binding site within this family, distinct from the ion-binding characteristics of other zinc transporters in the ZnT family. Combined with cellular experiments involving mutations at these critical residues for Mn2+ transport, the study revealed their indispensable role in Mn2+ transport and the maintenance of mitochondrial homeostasis.
|
|
| 
|
Structural basis and validation of key residues involved in SLC30A10-mediated Mn2+ transport.
Simultaneously, the research discovered that, in the presence of Mn2+, SLC30A10 exhibits symmetrical inward-facing (IF-IF) and asymmetrical inward-facing/outward-facing (IF-OF) conformations. Based on this, a model was proposed whereby the cooperative rotation of TM5, TM1, and TM2 facilitates the release of Mn2+ from the transmembrane binding site to the extracellular space. Through functional assays measuring cellular ROS, mitochondrial membrane potential, and ATP production, the team further confirmed that the HMNDYT1-associated hereditary mutation D40A disrupts SLC30A10's ability to bind and transport Mn2+ , leading to cell death upon Mn2+ overload. This suggests that the D40 residue may be a potential therapeutic target for HMNDYT1. Furthermore, mutation validation and complementation experiments conducted in SH-SY5Y neuronal cells provided preliminary insights into the impact of SLC30A10-mediated Mn2+ transport on cellular oxidative stress and mitochondrial homeostasis, and offered an exploration of the potential mechanisms underlying its neurotoxicity. This study provides the first systematic elucidation at the molecular level of the Mn2+ selectivity and conformational transition pathway of SLC30A10, offering a structural foundation for interpreting pathogenic mutations and screening for modulators relevant to HMNDYT1 and related neurological disorders.
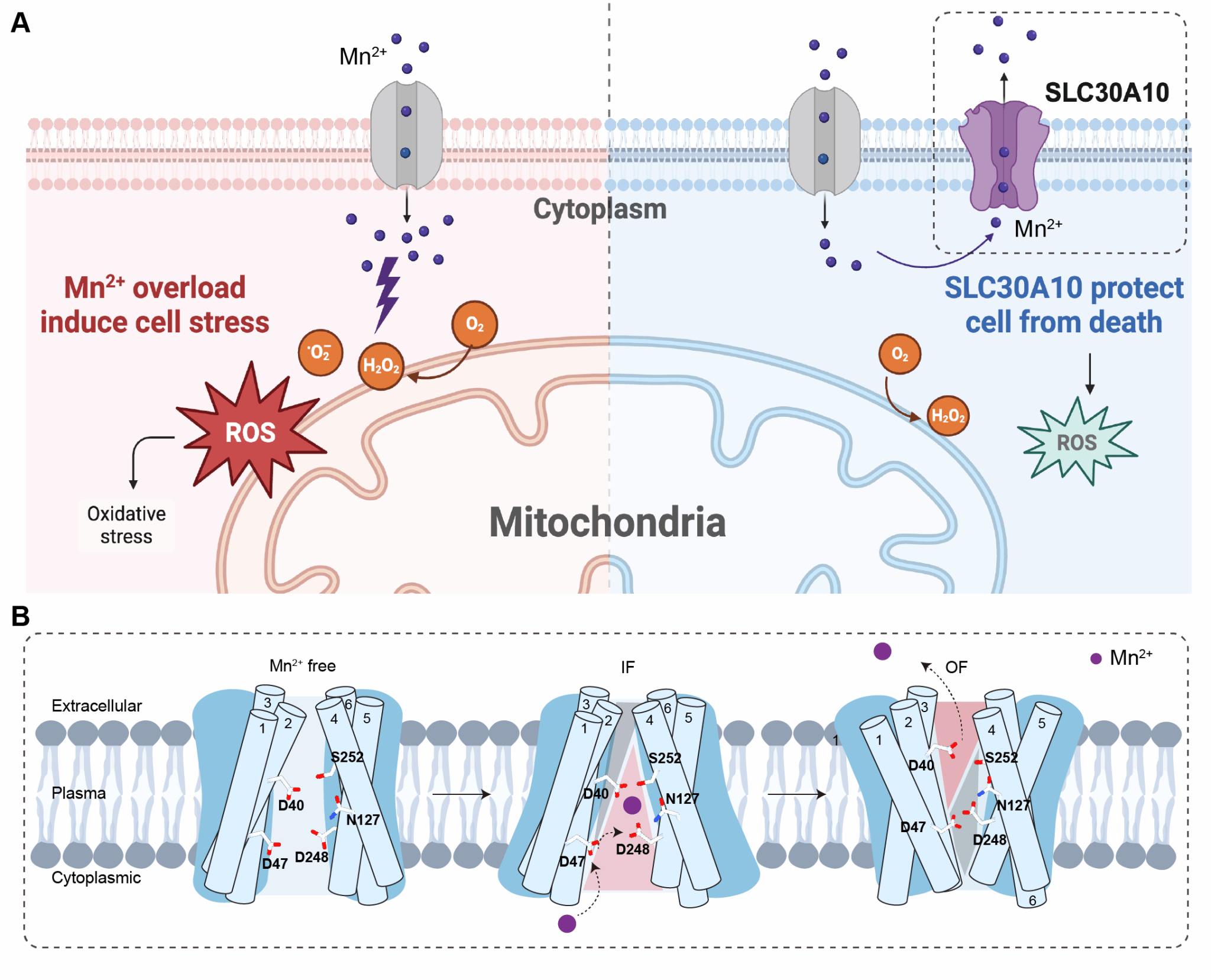
Schematic diagram illustrating the mechanism of manganese homeostasis regulation and transmembrane transport mediated by SLC30A10.
Researcher Yang Hanting from the Institute for Translational Brain Research, Fudan University, is the corresponding author of this paper. Dr Shen Xurui and doctoral student Zhang Jinlun, both from the same institute, are the co-first authors. This research was supported by funding from the Science and Technology Innovation 2030 -“ "Brain Science and Brain-Inspired Research" Major Project (Youth Program), the National Natural Science Foundation of China, and the China Postdoctoral Science Foundation.
Link to the full article: https://www.nature.com/articles/s41467-025-63616-7