Memory is not fixed. Fear memory offers a clear example: humans and animals must rapidly learn to remain vigilant after danger, a process essential for survival. Yet it is equally important for the brain to let go of fear once the environment becomes safe. Fear extinction is the classical model of this updating process: when an individual is repeatedly exposed to a previously threatening cue in the absence of harm, fear responses gradually diminish. This process is closely related to exposure-based, trauma-focused psychotherapy used in the clinic. In reality, however, extinction learning is often fragile and prone to relapse over time or when the context changes—one of the central challenges in the treatment of post-traumatic stress disorder (PTSD). Understanding why extinction occurs, and when it can be stabilized, is therefore a fundamental question in affective memory research.
For many years, researchers have focused on the small populations of excitatory glutamatergic neurons recruited by learning events—so-called memory engrams. More recently, inhibitory GABAergic interneurons have emerged as more than a passive background brake on excitation: they can help determine which neurons enter an engram, when an engram becomes susceptible to updating, and when it instead becomes stabilized. Notably, some interneurons release not only fast GABA but also slower signals such as neuropeptides. Because neuropeptides act over longer timescales and may diffuse more broadly, they offer a new window into how memory lability and stability are coordinated within the same circuit. Yet direct evidence for the molecular and temporal logic underlying this coordination has been lacking.

On March 31, 2026, a team led by Prof. Wei-Guang Li at our institute and Professor Tian-Le Xu at Shanghai Jiao Tong University School of Medicine published a research article in Nature Neuroscience entitled “Neuropeptide Y co-opts neuronal ensembles for memory lability and stability.” The study focuses on a class of GABAergic interneurons in the ventral hippocampal CA1 region (vCA1) that express neuropeptide Y (NPY). By integrating activity-dependent single-cell transcriptomics, in vivo fiber photometry, fluorescent neuropeptide sensors, optogenetics, and gene editing, the team dynamically tracked and causally manipulated NPY signaling during fear extinction.
They found that extinction training selectively recruits NPY neurons and progressively increases NPY release as training proceeds. More importantly, NPY signaling unfolds in clearly separable stages. An early, rapid increase in NPY facilitates the initiation of extinction, making fear memory more susceptible to updating. A later, sustained rise in NPY shapes the ultimate extent of extinction and is closely linked to whether the updated memory can be stably maintained. By linking the temporal dynamics of a neuropeptide to the transition between memory lability and stability, the study provides new molecular- and circuit-level insight into why extinction memories are often fragile, and when a more enduring “safety memory” may form.
The authors tested both the necessity and sufficiency of the NPY system using multi-level causal manipulations. Inhibiting NPY neurons during extinction training markedly impaired extinction, preventing fear responses from declining normally. In contrast, activating NPY neurons or locally supplying NPY in vCA1 accelerated the reduction in fear and improved later retrieval of extinction memory. The team further divided extinction into two partially separable phases: an early phase marked by a rapid drop in freezing, followed by a slower and more stable plateau. NPY interneurons proved especially important for the early phase—when their activity was suppressed, early extinction failed to start properly, which in turn disrupted the later stable phase.
Mechanistically, NPY interneurons release both fast GABA and the slower neuropeptide NPY. The study shows that these two signals serve distinct functions: fast GABA transmission is more critical for the initial formation of fear memory, whereas NPY signaling is especially important during extinction. In other words, the same NPY interneurons use two chemical signaling modes operating on different timescales to support memory encoding and subsequent updating within a single microcircuit.
NPY itself does not act through a single pathway. Instead, the authors found that it engages two receptor systems that influence distinct neuronal populations encoding fear versus extinction engrams. Specifically, NPY2R is mainly expressed in deep-layer vCA1 pyramidal neurons. Because it has a high affinity for NPY, it is preferentially engaged when NPY levels are still relatively low, thereby promoting memory lability and allowing fear responses to be more readily updated by extinction training. NPY1R, by contrast, is mainly expressed in superficial and middle pyramidal layers and has lower affinity, such that it is recruited more strongly only after NPY has accumulated. Its function appears to be to set a boundary on the updating process during later stages, thereby shaping the endpoint of extinction and helping the system return to a relatively stable state once updating is complete. The neuronal populations defined by these two receptors are spatially segregated with limited overlap, and NPY likely diffuses through the local circuit by volume transmission, sequentially influencing receptor-positive populations and thereby enabling staged regulation (Figure 1).
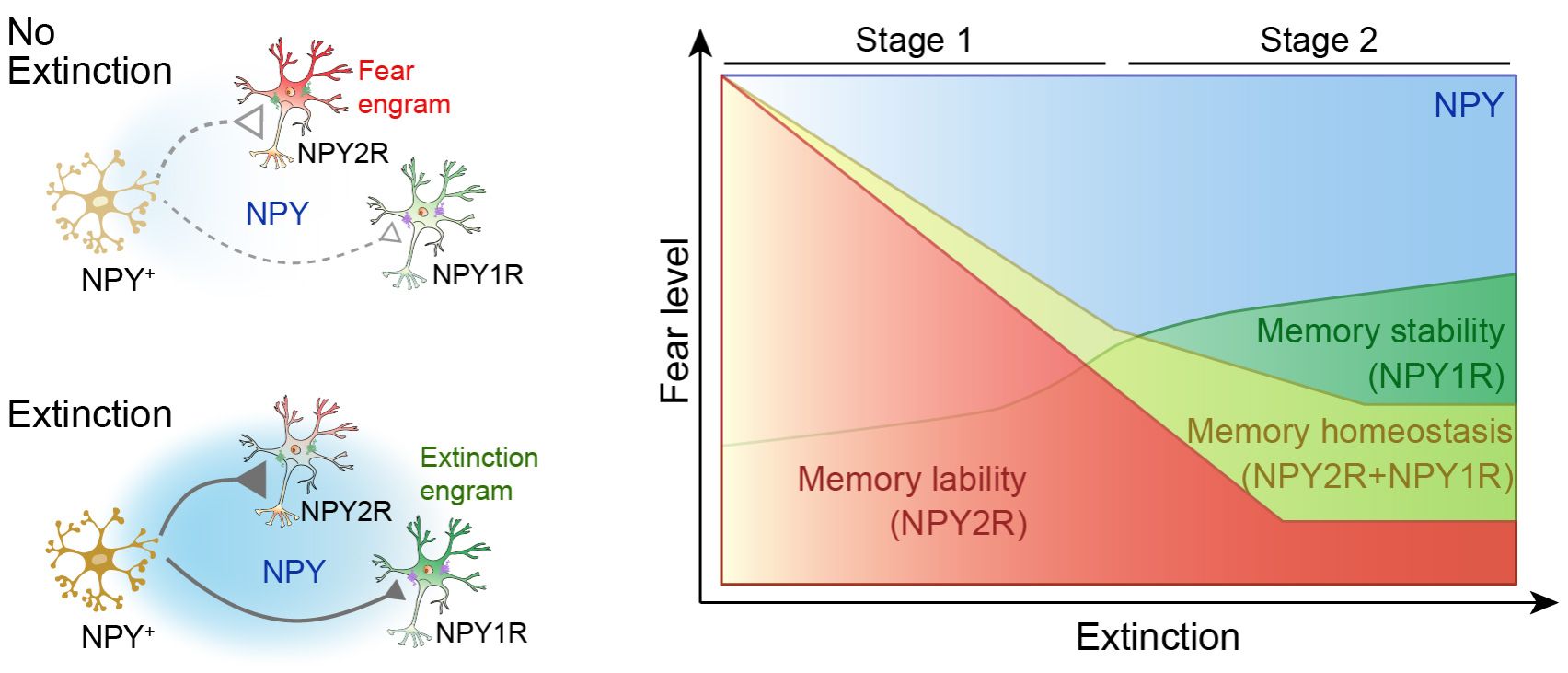
Figure 1. Schematic illustration of the molecular mechanism by which slow peptidergic signaling from NPY inhibitory interneurons in ventral hippocampal CA1 drives the dynamic updating of fear memory through distinct receptor-defined neuronal ensembles.
Overall, this work places the interneuron–neuropeptide–neuronal ensemble axis into a temporal framework and proposes that NPY regulates fear extinction in a staged manner: early on, it creates the conditions for a memory to become plastic and updateable; later, it helps set the endpoint of that update and promotes stable maintenance. The findings deepen our understanding of fear extinction and highlight the importance of timing in therapeutic intervention—suggesting that the same molecular signal may serve different functions at different stages. More broadly, the study offers a new conceptual route for enhancing the stability of extinction learning, reducing relapse of fear, and identifying more effective time windows to promote the formation and consolidation of safety memories.
The paper’s co-first authors are Dr. Yan-Jiao Wu, a former postdoctoral fellow at Shanghai Jiao Tong University School of Medicine and now a research associate professor at our institute; Dr. Xue Gu, assistant researcher; and PhD students Yalei Kong and Shuo Yang. The corresponding authors are Professor Wei-Guang Li from our institute and Professor Tian-Le Xu from Shanghai Jiao Tong University School of Medicine. Additional support came from Profs. Qian Li and Ju Huang at Shanghai Jiao Tong University School of Medicine, Yulong Li at the School of Life Sciences, Peking University, Lu-Yang Wang at the Department of Physiology, University of Toronto, and Miachael Xi Zhu at Shenzhen Bay Laboratory, among others. The study was supported by the Science and Technology Innovation 2030 – Brain Science and Brain-Inspired Research major program, the National Natural Science Foundation of China, and projects from multiple levels in Shanghai.
Article link: https://www.nature.com/articles/s41593-026-02235-x